Research - International Journal of Medical Research & Health Sciences ( 2021) Volume 10, Issue 10
Do Gastrointestinal Symptoms in COVID-19 Patients Correlate with Disease Severity?
Varun Malhotra1, Kashish Jafri2, Hena Dhir3 and Rahul Bhargava4*2Department of Community Health and Humanity, Memorial University, Newfoundland, Canada
3Casualty Medical Officer, Government Medical College, Chandigarh, India
4Department of Ophthalmology, GS Medical College, Pilkhuwa, UP, India
Rahul Bhargava, Department of Ophthalmology, GS Medical College, Pilkhuwa, UP, India, Email: brahul_2371@yahoo.co.in
Received: 28-Sep-2021 Accepted Date: Oct 22, 2021 ; Published: 29-Oct-2021
Abstract
Background: Apart from the respiratory tract symptoms, patients with COVID-19 infection may be either asymptomatic or present with Gastrointestinal (GI) symptoms. However, it is not known whether the presence of GI symptoms has any prognostic value or their presence is associated with poor disease outcomes in COVID-19 patients. Objective: To determine the prevalence of GI symptoms and their correlation with disease severity and outcome in patients with COVID-19. Methods: A hospital-based controlled study. Results: The number of valid cases was 444 with a mean age of 46 ± 18.6 (range, 18-80 years). One hundred and twenty (27%) patients presented with GI symptoms alone and 12% of patients were asymptomatic. Throughout illness, 304 (68.4%) patients developed GI symptoms. Diarrhea was the most common GI symptom (34.2%), followed by loss of appetite (33%) and loss of taste (28.8%), and tongue ulcers (14.6%). There was a significant difference (Chi-square tests, p=0.006) in COVID-19 severity between patients with and without GI symptoms. Oxygen supplementation was required in 174 (39.2%) patients, mechanical ventilation was required in 34 (7.7%). The mean stay in the Intensive Care Unit (ICU) was 13.5 ± 7.4days. The mean duration of oxygen therapy was 10.4 ± 6.4 days and the mean duration of mechanical ventilation was 10.8±4.7days. There was a significant difference in outcome between patients with and without GI symptoms (Chi-square tests, p=0.033). Conclusion: The presence of GI symptoms does correlate with COVID-19 disease severity. However, their impact on clinical outcome and mortality needs further evaluation.
Keywords
COVID-19, Gastrointestinal manifestation, Disease severity, Clinical outcome
Introduction
The latest pandemic to affect the world is the novel coronavirus infection 2019, now known as Corona Virus Disease 2019 (COVID-19). The source of infection was initially traced to Hubei province in Wuhan city of China; the infection thereafter blew out exponentially and disrupted the lives of billions of people and compromised hundreds of economies around the world within a few months [1].
Previous pandemic Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV) and Middle Eastern Respiratory Syndrome Coronavirus (MERS-CoV), respectively, were predominantly, respiratory tract infections [2]. Likewise, acute respiratory symptoms like cough, nasal congestion, high-grade fever, dyspnoea, pneumonia with infiltrations and Acute Respiratory Distress Syndrome (ARDS) have been predominantly reported in the ongoing COVID-19 pandemic [3,4]. However, it has been noticed that a considerable number of patients are either asymptomatic or may present with Gastrointestinal (GI) manifestations at the outset or may develop GI symptoms during the disease [5,6].
The commonly reported gastrointestinal symptoms in COVID-19 patients are nausea, vomiting, diarrhea, pain abdomen, ageusia, anosmia, and loss of appetite [7,8]. Apart from these symptoms, some patients may experience severe GI disease in the form of GI bleed, acute pancreatitis, bowel perforation, and liver injury [9,10]. In the present series, additionally, tongue ulcers were observed in 14.3% of cases.
In a single-center cohort study in Italy, 69% of COVID-19 patients had at least one GI symptom at diagnosis. However, patients with GI symptoms, particularly diarrhea had a lower mortality rate [11]. In another study from the same country, the prevalence of GI symptoms in COVID-19 patients at admission was 8.9% and their presence was associated with a milder course of disease [12]. On the contrary, a single-center retrospective cohort study found that patients with GI symptoms (n=359) had a higher ARDS and subsequent need for mechanical ventilation as compared to those without GI symptoms (n=718). However, the mortality rates were comparable [13].
Studies have documented that SARS-CoV-1 and SARS-CoV-2 enter into human host cells through Angiotensin- Converting Enzyme 2 (ACE-2) receptors; these proteins are found in pneumocytes, small intestine lining epithelium, renal proximal tubular cells, endothelial cells, and smooth muscle cells of the arteries. ACE has been reported to control intestinal inflammation and this may be one of the mechanisms by which SARS-COV-2 causes diarrhea and other GI symptoms [14,15].
The focus for COVID-19 tracing and subsequent prognostication has largely been placed on respiratory symptomatology and the extent of lung involvement, respectively. The presence of GI manifestations has been overlooked to a large extent. Patients with these atypical presentations as mentioned are often missed or misdiagnosed till they start manifesting respiratory signs and symptoms at a later stage. Thus, and delay in diagnosis may potentially expose close contacts like family members and health care workers to the COVID-19 virus transmission by another potential (fecal-oral) or unknown routes [16].
Although there is no universally accepted consensus on the treatment of the COVID-19 disease yet, a better understanding of the mechanism of virus transmission could be prudent for better management shortly as new disease variants of SARS-COV-2 have been reported in some countries. Second, a better understanding of atypical presentations of COVID-19 may decrease the symptom-to-admission time and reduce the exposure to first-line health care workers.
The present study aimed to determine the prevalence of GI manifestations in COVID-19 patients admitted to a tertiary COVID center and assess their correlation with the severity of the disease.
Methods
A hospital-based controlled study was conducted at a tertiary care teaching hospital in the Indian sub-continent from July to November 2020. The aim of the study aimed was to evaluate the prevalence of gastrointestinal manifestations in patients affected by COVID-19 and whether any correlation exists between GI symptoms and COVID-19 disease severity and disease outcome. Written informed consent was obtained from all patients based on tenets of the declaration of Helsinki.
Inclusion Criteria
All patients who test positive for SARS-CoV-2 by RT-PCR or GeneXpert were included in the study.
Exclusion Criteria
Patients less than 15 years were excluded from the study. A patient who received anti-viral treatment during the first week of the presentation was also excluded from the study.
The primary outcome measures were to determine the prevalence of gastrointestinal symptoms in COVID-19 patients and their correlation with the severity of COVID-19 infection. The secondary outcome was to evaluate pancreatic enzyme elevation among COVID-19 patients and to compare the prevalence of GI symptoms in patients with and without respiratory symptoms. The presence of GI symptoms was also evaluated in terms of hospital stay, ICU stays, need for ventilator support, and prognostic value.
A detailed history was taken regarding symptoms such as fever, cough, sore throat, shortness of breath, chest tightness, chest pain and rhinorrhea, GI symptoms such as nausea, vomiting, diarrhea, pain abdomen or discomfort, reflux, heartburn, bloating, dyspepsia, loss of appetite, hematemesis, melena, loss of taste and smell were inquired and duration of these symptoms was recorded. All patients were also inquired about existing comorbidities. Clinical examination of the patients was retrieved from the physician in charge of the patient and file records.
These patients were divided into two groups:
Group I: Patients who developed GI symptoms (at onset or during hospital stay)
Group II: Patients who did not develop any GI symptoms.
All patients had routine blood tests such as Complete Blood Count (CBC), Serum Electrolytes (SE), Renal Function Test (RFT), lipid profile, amylase, lipase, calcium, phosphorus, stool, and saliva RT-PCR and stool routine examination. Stool RT-PCR was done again for all patients 2 weeks after the discharge. Other investigations such as ultrasound abdomen, chest X-ray, CT scan were carried out as per the clinical needs of the patient.
All patients were classified according to the clinical severity of their basic disease. The index of severity was adopted from the definition by the Ministry of health and family welfare of India as published on their website on 13.6.2020 [17]. All patients were managed as per the institute protocol based on a multi-disciplinary team decision on a case-tocase basis.
Statistics
Statistical analysis was performed using IBM, SPSS Statistics version 26 (IBM Inc.). Data are expressed as mean and Standard Deviation (SD) unless otherwise stated. p-value less than 0.05 was considered statistically significant. Continuous variables were compared using a t-test. Wherever necessary, the student t-test was altered to compare unequal variances. The assumptions of performing t-tests were met. The independent-samples t-test was used to determine if a difference exists between the means of two independent groups on a continuous dependent variable (gender and age, presence and absence of gastrointestinal symptoms, and biochemical investigations). Chi-square tests were used for proportions. A one-way repeated measures ANOVA was conducted to determine whether there was a statistically significant difference in serum enzymes between grades of severity of COVID-19 infection. There were no outliers on visual inspection of boxplots. The data were normally distributed at each time point, as assessed by the Shapiro-Wilk test (p>0.05), respectively. The assumption of sphericity was violated, as assessed by Mauchly’s test of sphericity, so a Greenhouse and Geisser correction was applied. Post hoc analysis was done with a Bonferroni adjustment to see the point of time when a change in test parameters occurred after the intervention. Point biserial correlation was done to see the association between presence/absence of GI symptoms and ICU stay, Oxygen therapy, and mechanical ventilation duration.
Results
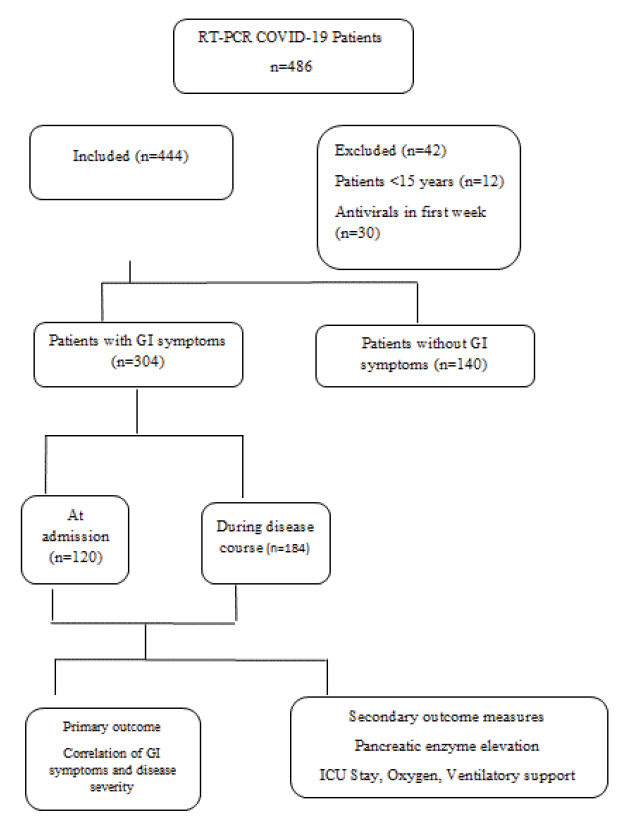
A total of four hundred and forty-four patients were included for analysis. The flow chart showing patient enrolment and allocation is depicted in Figure 1.
The mean age of patients was 46 ± 18.6 (range, 18-80 years). Table 1 mentions the clinical characteristics of study participants. There were 280 (63%) males with a male-female ratio of 1.7:1. The mean age of males was 43.8 ± 16.1 years, and the mean age of females was 39.2 ± 17.3 years, respectively (p=0.048). The mean age of patients without GI symptoms was 41.2 ± 16.1 and with GI symptoms was 43.9 ± 17.8 years, respectively (independent t-test, p=0.271). Gastrointestinal symptoms did not significantly differ between males and females (Chi-square test, p=0.460).
| Patient characteristic | All patients (444) | GI symptoms (304) | No GI symptoms (140) |
|---|---|---|---|
| Age | 44.6 ± 15.4 | 43.9 ± 17.8 | 41.2 ± 16.1 |
| Comorbidities (n, %) | 62 (14) | 32 (10.5) | 30 (21.4) |
| Diabetes | 102 (23) | 62 (20.4) | 40 (28.6) |
| Hypertension | 172 (38.7) | 102 (33.6) | 70 (50) |
| CVD | 40 (9) | 24 (7.9) | 16 (11.4) |
| AST(U/L) | 44.2 ± 18.6 | 56.4 ± 20.6 | 40.8 ± 20.8 |
| ALT(U/L) | 34 ± 19.8 | 40.6 ± 22.4 | 32.8 ± 20.2 |
| CRP (mg/dl) | 104 ± 60.6 | 90 ± 64.8 | 96 ± 66.8 |
| D-dimer (nmol) | 1980 ± 4876 | 1768 ± 5200 | 1684 ± 4600 |
| Hospital stays (days) | 12.4 ± 4.6 | 13.7 ± 5.2 | 11.6 ± 6.2 |
| ICU stay (days) | 6.4 ± 4.8 | 5.4 ± 5.2 | 4.4 ± 5.5 |
| Abbreviations: AST (Aspartate Amino Transferase), ALT (Alanine Amino Transferase), CRP (C-Reactive Protein), ICU (Intensive Care Unit) | |||
In our study cohort, 180 (40.5%) COVID-19 positive patients were health care workers. None of the female patients were pregnant at the time of initial diagnosis. The mean duration of hospital stay was 13.7 ± 5.2 days. On presentation, fever was the most common symptom present in 292 (65.8%) patients and 18 (4.1%) patients were asymptomatic contacts of COVID-19 patients.
At least one gastrointestinal symptom was found to be present in 27% of study participants (120 patients) as a presenting complaint; out of these 92 patients had GI symptoms in combination with other symptoms. During illness, overall, 304 (68.4%) patients developed GI symptoms. Sixty patients had only GI manifestations without any respiratory symptoms. About 12% of patients have neither GI nor respiratory symptoms. These patients either had isolated fever, chest pain or were asymptomatic throughout the course.
Throughout illness, fever was the most common symptom (78%), followed by cough (52%) and shortness of breath (48%). Among GI manifestations (Table 2), diarrhea was the most common symptom (34.2%), followed by loss of appetite (33%), loss of taste (28.8%), and tongue ulcers (14.3%). Of the 444 patients, 124 (27.9%) patients were diabetic and 122 (27.4%) were hypertensive.
| GI Symptom | N (%) |
|---|---|
| Diarrhea | 104 (34.2) |
| Loss of appetite | 100 (33) |
| Loss of taste | 86 (28.8) |
| Nausea | 61 (20) |
| Tongue ulcers | 43 (14.3) |
| Melena | 6 (1.9) |
The patients were also inquired specifically about details of their pre-existing GI morbidities. GI pathologies were present in 7.7% of the patients. Twelve (2.7%) patients had Chronic Liver Disease (CLD) (Alcohol-related- 8, Hepatitis C related-2 and Hepatitis B related-1). Ten patients presented with acute pancreatitis. Four patients had alcohol-related acute pancreatitis, 6 patients had a history of choledocholithiasis, 6 patients had asymptomatic gall stone disease and 5 patients had melena.
GI Symptoms and COVID-19 Severity
Of the 444 enrolled patients, 270 (60.8%) had the mild respiratory disease while 70 patients had severe disease. Nine of the 70 patients with the severe disease also had septic shock. The severity of the disease was compared among the patients with or without GI symptoms. There was a significant difference (Chi-square tests, p=0.006) in COVID-19 severity between patients with and without GI symptoms (Table 3).
| COVID-19 Severity Grade | GI Symptoms (n=304) | No GI Symptoms (n=140) | p-value |
|---|---|---|---|
| Mild | 172 (56.6%) | 98 (70%) | 0.006* |
| Moderate | 68 (22.4%) | 36 (25.7%) | |
| Severe | 64 (21.0%) | 6 (4.3%) | |
| Abbreviations: GI: Gastro-Intestinal; *Chi-Square Test | |||
Most patients with diarrhea required only supportive treatment. Metronidazole was given to only 10 patients. One patient received the combination of ofloxacin and ornidazole following which diarrhea subsided but resumed after the drug was stopped. Twelve patients had GI bleeding, 8 of which required Endoscopic Variceal Ligation (EVL). GI bleed in the other two patients stopped with conservative management. Symptomatic treatment and supportive care were prescribed for patients with other GI symptoms.
Out of 444 patients, oxygen supplementation (Table 4) was required in 174 (39.2%) patients, ventilation support was required in 34 (7.7%). The mean stay in the Intensive Care Unit (ICU) was 13.5 ± 7.4 days. The mean duration of oxygen therapy was 10.4 ± 6.4 days and the mean duration of mechanical ventilation was 10.8 ± 4.7 days.
| Parameter (n, %) | GI Symptoms present | GI symptoms Absent | p-value |
|---|---|---|---|
| Oxygen Supplementation | 132 (43.4) | 44 (14.5) | 0.06 |
| Ventilator support | 30 (9.8) | 4 | 0.05 |
| Dialysis (Renal support) | 16 | 12 | 0.379 |
| Vasopressor support (CVS support) | 10 | 8 | 0.468 |
| ICU Stay | 54 | 4 | 0.01 |
| Abbreviations: ICU: Intensive Care Unit, CVS: Cardio-Vascular System | |||
In our study cohort, amylase was raised in 94 (21.17%) patients and lipase was raised in 156 (34.68 %) patients. Mean amylase and lipase levels in our patients were 82.8 ± 68 U/L (Standard range: 28-100 U/L) and 63.3 ± 70 U/L (Standard range: 13 U/L-60 U/L) respectively.
Elevated liver enzymes were found to be present in many COVID-19 patients. Aspartate Aminotransferase (AST) was found to be raised in 178 (40.1%) patients and elevated Alanine Aminotransferase (ALT) was found in 214 (48.2%) patients. The mean level of AST was 46 ± 37 U/L (standard range: 2 U/L-41 U/L) and that of ALT was 52 ± 50 (standard range: 2 U/L-41 U/L).
There was a significant difference in outcome between patients with and without GI symptoms (Chi-square tests, p=0.033).
Discussion
A prospective, interventional cross-sectional study was conducted at a tertiary care cum COVID-19 regional facility in the northern part of the sub-continent. This study evaluated gastrointestinal symptoms in patients who were infected with COVID-19 between March to September 2020.
Although a lower respiratory tract infection transmitted through respiratory droplets, gastrointestinal symptoms are increasingly been reported across different studies worldwide and disease many patients with COVID-19 may present with gastrointestinal symptoms perse [18,19].
Our study revealed that the prevalence of gastrointestinal symptoms at presentation was 27%. The overall prevalence of gastrointestinal symptoms during illness was 68.5%. At presentation, the most common GI symptom was nausea 8.1%; however, diarrhea 30.2% was the most common GI symptom during the illness.
In a study in China (Wuhan), retrospective analysis of 1141 COVID-19 patients over 7 weeks revealed that 16% of the patient’s presented with gastrointestinal symptoms. Out of these (n=183), the most common presenting symptoms were loss of appetite, followed by nausea and vomiting in 66.6% of the patients, diarrhea in 37%, and abdominal pain in 25% cases, respectively. In this study, mild elevation of liver enzymes (AST and ALT) was observed but renal functions were unaffected [20]. In our study, AST and ALT were deranged in 128 (42.1%) and 158 (51.9%) patients with gastrointestinal symptoms, respectively.
Mao and co-authors conducted a meta-analysis of 35 studies involving 6686 COVID-19 patients. Gastrointestinal symptoms were reported in 29 studies (n=6064). The authors found that the pooled prevalence of GI symptoms was 15% (95% CI, 10-21); nausea, vomiting, diarrhea, and anorexia were the most frequent symptoms. About 10% of patients presented with gastrointestinal symptoms without respiratory features. In our study, 60 (13.5%) patients had only GI symptoms without respiratory features which were comparable to the results of this meta-analysis. On subgroup analysis, the authors found that patients with severe COVID-19 had higher rates of abnormal liver function including increased ALT (1.89 [95% CI, 1.3-2.76]; p=0.0009) and increased AST (3.08 [95% CI, 2.1-4.4]; p<0.00001) compared with those with the non-severe disease. In our study, the point prevalence of deranged liver enzymes was higher. Second, the height of liver enzyme elevation did not significantly (p=0.700 and 0.356 for AST and ALT, respectively) correlate with the severity of COVID-19 infection. This could be explained by the fact that the sample size in our study was small (304 versus 6064) and thereby increasing the probability of overestimation (Type II error) [8].
A cross-sectional descriptive study from Hubei province, China, in 204 COVID-19 patients found that gastrointestinal symptoms were present in 99 (48.5%) cases. Of these, the GI symptoms were anorexia in 83 (83.8%), diarrhea in 29 (29.3%), vomiting in 8 (8.1%), and abdominal pain in 4 (4.0%), respectively; some patients presented with multiple symptoms. Seven patients only had GI symptoms without any respiratory symptoms [7].
In contrast to the above-mentioned study, a study of 1099 patients (China) found that the most common presenting symptoms at admission were fever (43.8%) and cough (67.8%). However, gastrointestinal symptoms were minimal; nausea or vomiting in 5% and diarrhea in 3.8% of patients [21].
Thus, it appears that GI involvement in COVID-19 patients and liver dysfunction often co-exists. Some researchers think that liver dysfunction is synonymous with disease severity upon presentation. However, it cannot be said whether liver dysfunction is due to the effect of viral infection per se or due to treatment modalities. This is reflected in gross variation in the prevalence of liver enzyme abnormalities across studies.
In one study, total bilirubin, AST, and ALT were elevated in 10%, 21%, and 22% of patients, respectively [21]. In other studies, liver enzyme abnormalities range from 16%-53%. The prevalence of liver enzyme elevation in our study falls within the upper limit of the reported prevalence [22]. These observations may be critically important as drugs like Remdesivir, often prescribed for COVID pneumonia, often cause hepatic dysfunction per se.
In retrospective single-center studies in Italy, the presence of GI symptoms, particularly diarrhea was associated with low mortality and a milder form of respiratory illness [11,12]. In our study, COVID-19 patients with diarrhea were associated with a significantly higher prevalence of ARDS and respiratory complications. However, our findings were substantiated in another study by Chen, et al.; the authors found that in patients with GI symptoms, there was a significantly higher requirement of oxygen supplementation and ventilator support due to ARDS but the mortality rates were comparable to those without GI involvement [13].
Patients with pre-existing GI diseases may be at higher risk of COVID-19 infection and severity, due to immunodeficiency [23]. In our study, no correlation was observed between patients with liver disease and COVID-19 severity (p=0.345). These findings were consistent with other published studies where no statistically significant association was found between underlying chronic liver disease and severity of COVID-19 disease [24]. However, the study by Guan, et al. found that 2.1% of patients having underlying hepatitis B were associated with more severe COVID-19 disease [21].
Ramachandran, et al. retrospectively analyzed 150 hospitalized COVID-19 patients at a tertiary care center. The study aimed to assess if GI symptoms had prognostication value in hospitalized patients with COVID-19. Thirty-one (20.6%) patients with 1 or more of the GI symptoms were compared with 119 COVID-19 patients without GI symptoms which served as controls. The authors did not find any significant difference in comorbidities and laboratory findings. The mortality rate did not significantly differ between cases and controls (p=0.680). The secondary clinical outcomes, including the length of stay (p=0.870) and need for mechanical ventilation (p=0.820), were comparable between the two groups [25]. In our study, the duration of hospital stays (p=0.761) and need for mechanical ventilation (p=0.053) were comparable between the two groups. However, there was a significant difference in the duration of ICU stay (0.001) and the outcome. The mortality rate was significantly higher (p=0.033) in patients with GI symptoms.
The limitations of the present study were a small sample size (n=39) thereby increasing the likelihood of type II error and consequently overestimation. Second, the selection of patients was not random, potentially leading to selection bias. The study design although prospective and interventional had a short follow-up of patients, making associations difficult to interpret.
Conclusion
In conclusion, patients with COVID-19 frequently present with gastrointestinal symptoms and these may be the only presenting feature of COVID-19 infection in some patients. The presence of GI symptoms appears to correlate with COVID-19 disease severity but whether they have has any impact on mortality remains debatable and needs further evaluation in large multicentric studies.
Declarations
Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Acknowledgments
We are thankful to www.indianmedicalststs.com for statistical analysis.
Financial Interests
We do not have any financial interests.
References
- World Health Organization. "Novel Coronavirus (2019-nCoV): Situation report, 1." 2020.
- Noor, Aziz Ullah, et al. "Epidemiology of Covid-19 Pandemic: Recovery and mortality ratio around the globe." Pakistan Journal of Medical Sciences, Vol. 36, No. COVID19-S4, 2020, pp. S79-84.
- Assiri, Abdullah, et al. "Epidemiological, demographic, and clinical characteristics of 47 cases of Middle East respiratory syndrome coronavirus disease from Saudi Arabia: A descriptive study." The Lancet Infectious Diseases, Vol. 13, No. 9, 2013, pp. 752-61.
- Mackay, Ian M., and Katherine E. Arden. "MERS coronavirus: Diagnostics, epidemiology and transmission." Virology Journal, Vol. 12, No. 1, 2015, pp. 1-21.
- Samanta, Jayanta, et al. "2019 novel coronavirus infection: Gastrointestinal manifestations." Journal of Digestive Endoscopy, Vol. 11, No. 1, 2020, pp. 13-18.
- Smyk, Wiktor, et al. "COVID‐19: Focus on the lungs but do not forget the gastrointestinal tract." European Journal of Clinical Investigation, Vol. 50, No. 9, 2020, p. e13276.
- Pan, Lei, et al. "Clinical characteristics of COVID-19 patients with digestive symptoms in Hubei, China: A descriptive, cross-sectional, multicenter study." The American Journal of Gastroenterology, Vol. 115, 2020, pp. 763-73.
- Mao, Ren, et al. "Manifestations and prognosis of gastrointestinal and liver involvement in patients with COVID-19: A systematic review and meta-analysis." The Lancet Gastroenterology and Hepatology, Vol. 5, No. 7, 2020, pp. 667-78.
- Gulen, Muge, and Salim Satar. "Uncommon presentation of COVID-19: Gastrointestinal bleeding." Clinics and Research in Hepatology and Gastroenterology, Vol. 44, No. 4, 2020, pp. e72-76.
- Hadi, Amer, et al. "Coronavirus disease-19 (COVID-19) associated with severe acute pancreatitis: Case report on three family members." Pancreatology, Vol. 20, No. 4, 2020, pp. 665-67.
- Schettino, Mario, et al. "Clinical characteristics of COVID-19 patients with gastrointestinal symptoms in Northern Italy: A single-center cohort study." Official Journal of the American College of Gastroenterology| ACG, Vol. 116, No. 2, 2021, pp. 306-10.
- Papa, Alfredo, et al. "Gastrointestinal symptoms and digestive comorbidities in an Italian cohort of patients with COVID-19." European Review for Medical and Pharmacological Sciences, Vol. 24, No. 13, 2020, pp. 7506-11.
- Chen, Rong, et al. "Gastrointestinal symptoms associated with unfavorable prognosis of COVID-19 patients: A retrospective study." Frontiers in Medicine, Vol. 7, 2020.
- Hoffmann, Markus, et al. "SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor." Cell, Vol. 181, No. 2, 2020, pp. 271-80.
- Deshmukh, Vishwajit, et al. "COVID-19: A conundrum to decipher." 2020.
- Medema, Gertjan, et al. "Presence of SARS-Coronavirus-2 RNA in sewage and correlation with reported COVID-19 prevalence in the early stage of the epidemic in the Netherlands." Environmental Science and Technology Letters, Vol.7, No. 7, 2020, pp. 511-16.
- Clinical management protocol: COVID‐19. "Government of India Ministry of Health and Family Welfare Directorate General of Health Services (EMR Division)." 2020.
- Wu, Fan, et al. "A new coronavirus associated with human respiratory disease in China." Nature, Vol. 579, No. 7798, 2020, pp. 265-69.
- Yeo, Charleen, Sanghvi Kaushal, and Danson Yeo. "Enteric involvement of coronaviruses: Is faecal-oral transmission of SARS-CoV-2 possible?" The Lancet Gastroenterology and Hepatology, Vol. 5, No. 4, 2020, pp. 335-37.
- Luo, Shihua, Xiaochun Zhang, and Haibo Xu. "Don't overlook digestive symptoms in patients with 2019 novel Coronavirus Disease (COVID-19)." Clinical Gastroenterology and Hepatology, Vol. 18, No. 7, 2020, pp. 1636-37.
- Guan, Wei-jie, et al. "Clinical characteristics of coronavirus disease 2019 in China." New England Journal of Medicine, Vol. 382, No. 18, 2020, pp. 1708-20.
- Chen, Nanshan, et al. "Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study." The Lancet, Vol. 395, No. 10223, 2020, pp. 507-13.
- Chen, L., et al. "Analysis of clinical features of 29 patients with 2019 novel coronavirus pneumonia." Chinese Journal of Tuberculosis and Respiratory Diseases, Vol. 43, 2020, p. E005.
- Arentz, Matt, et al. "Characteristics and outcomes of 21 critically ill patients with COVID-19 in Washington State." JAMA, Vol. 323, No. 16, 2020, pp. 1612-14.
- Ramachandran, Preethi, et al. "Gastrointestinal symptoms and outcomes in hospitalized coronavirus disease 2019 patients." Digestive Diseases, Vol. 38, No. 5, 2020, pp. 373-79.