Research - International Journal of Medical Research & Health Sciences ( 2022) Volume 11, Issue 4
Prosthetic Valve Thrombosis: Literature Review and Two Case Reports
Kotryna Druteikaite1, Simona Berontaite1*, Gintare Valteryte2 and Gediminas Jarusevicius2,32Department of Cardiology, Lithuanian University of Health Sciences Kaunas Clinics, Kaunas, Lithuania
3Cardiology Institute, Lithuanian University of Health Sciences, Kaunas, Lithuania
Simona Berontaite, Faculty of Medicine, Academy of Medicine, Lithuanian University of Health Sciences, Kaunas, Lithuania, Email: simun147@gmail.com
Received: 09-Apr-2022, Manuscript No. ijmrhs-22-60184; Editor assigned: 10-Apr-2022, Pre QC No. ijmrhs-22-60184 (PQ); Reviewed: 20-Apr-2022, QC No. ijmrhs-22-60184 (Q); Revised: 20-Apr-2022, Manuscript No. ijmrhs-22-60184 (R); Published: 30-Apr-2022
Abstract
Prosthetic heart valve thrombosis is one of the most dangerous prosthetic valve complications. Proper monitoring and management of these patients help to prevent this complication. Fluoroscopy is advantageous in cases of thrombosis to assess the function of the prosthetic valve by measuring the opening and closing angles. We describe two cases of aortic mechanical valve thrombosis with different mechanisms of thrombus formation. The first case was a 48 years old woman admitted to the hospital because of shortness of breath during minimal exertion and significantly reduced exercise tolerance. Due to rheumatic heart disease, the patient underwent aortic and mitral mechanical prosthesis and has been using warfarin in therapeutic norms. During echocardioscopy aortic prosthesis obstruction and severe tricuspid valve regurgitation were observed. The patient was scheduled for aortic root and TV prosthesis surgery. The second patient also had an aortic mechanical valve due to severe aortic stenosis caused by rheumatism and presented with organizing pneumonia and progressing respiratory failure as complications of the COVID-19 infection and was admitted with dyspnea, cough and weakness. Aortic prosthetic valve thrombosis was diagnosed despite optimal treatment and therapeutic INR.
Keywords
Prosthetic valve thrombosis, Mechanical prosthesis, Aortic valve
Introduction
Prosthetic heart Valve Thrombosis (PVT) is an uncommon but usually life-threatening condition. PVT is a cause of valve prosthesis dysfunction and a complication of valve replacement surgery, occurring in any part of the heart valve prosthesis [1]. The exact rate of valve thrombosis is not known, but some authors estimate that incidence ranges from 0.5% to 6% per patient per year in the aortic and mitral position and up to 20% in the tricuspid position [2]. The main factors influencing the development of PVT are inappropriate treatment by anticoagulants, thrombogenicity of the valve, and hemodynamics of the transprosthetic blood flow [3]. Although thrombus formation is less frequent with new surgical techniques, the hemodynamic and physical properties of mechanical valves remain thrombogenic [4]. The purpose of this article is to review the epidemiology, clinical features, diagnosis, and updated management according to the current guidelines of PVT.
Literature Review
Indications for Aortic Valve Replacement, Prosthetic Valve Thrombosis Etiology, and Risk Factors
In Europe alone, more than 13 million people are diagnosed with Valvular Heart Disease (VHD) each year, and 100 million worldwide [5]. Aortic Valve (AV) disease is the most common among valvular conditions (44,3% of all VHD) and Surgical Aortic Valve Replacement (SAVR) or Transcatheter Aortic Valve Implantation (TAVI) is the main treatment for this condition [6,7]. Worldwide the number of AV replacements in 2003 was 290,000 and by 2050 is predicted to be 850,000 [8]. According to the latest ESC/EACTS guidelines, indications for AV replacement are severe symptomatic aortic regurgitation or asymptomatic aortic regurgitation in patients with Left Ventricular End- Systolic Diameter (LVESD) >50 mm or LVESD >25 mm/m2 Body Surface Area (BSA) in patients with small body size or resting Left Ventricular Ejection Fraction (LVEF) ≤ 50%. In the case of the aortic root or tubular ascending aortic aneurysm indications for surgery are Marfan syndrome who have aortic root disease with a maximal ascending aortic diameter ≥ 50 mm or aortic dilation, if performed in experienced centers and durable results are expected. In addition to, AV replacement is recommended in symptomatic patients with severe, high-gradient aortic stenosis or symptomatic severe low-flow, low-gradient aortic stenosis with LVEF <50% and evidence of flow (contractile) reserve [9]. Each surgery has its risks and one of the most common complications in valve replacement is PVT. Prosthetic valve dysfunction depends on the valve that has been implanted and on the procedure (SAVR or TAVI). Usually, the suitable valve is selected based on the following risk factors: anticoagulant-related bleeding and valve deterioration. Antithrombotic therapy slows down but does not eliminate the risk of PVT, which also depends on the patient’s lifestyle, metabolic profile, or inflammatory status, especially in the case of TAVI [10]. Predisposing factors for thrombosis include a high body mass index, male sex, bicuspid AV, and a large aortic root requiring a large-sized prosthesis [11]. There are several described mechanisms causing PVT, one of them is surface-induced thrombosis, which is well described in mechanical valves and other medical devices. The exposure of valve prostheses to blood leads to a thrombogenic process that results in the formation of thrombin, platelet activation, and the formation of platelet-fibrin networks on the surface of the prosthesis. Transcatheter aortic valves are also associated with pathological processes thromboembolic events commonly found in the first 3 months after the procedure. One hypothesis is that, because the original valve is not removed during TAVI but left in place, the leaflets of native valves still contain high levels of tissue factor, which enhances platelet activation [12].
Mechanical Aortic Valve Thrombosis
Mechanical AV replacement is recommended for patients who are younger than 60 years and have no contraindications to long-term anticoagulation [13]. Mechanical AV prostheses are characterized by their robust structure and long service life. Monoleaflet valves are composed of a single disc with an opening angle relative to the valve annulus ranging from 60° to 80°, resulting in 2 different sized orifices. Bileaflet valves consist of 2 semilunar discs, the opening angle of the discs varies from 75° to 90° and the open valve consists of 3 orifices [14]. When a mechanical valve is implanted, patients are at high risk of stroke and valve thrombosis and require long-term anticoagulant treatment. The risk of PVT is higher for mechanical mitral valves (0.9% of cases per year) than for mechanical AV (0.5% of cases per year). Longterm anticoagulation reduces the annual risk of PVT from 1.8% to 0.4% [15]. PVT is a serious complication occurring in 0.5%-8% of mechanical mitral and AV prostheses [16]. Thrombus formation is more frequently observed in the mechanical valve than in the bioprosthetic, presumably due to inadequate anticoagulant use, and is associated with Atrial Fibrillation (AFib) and poor cardiac function [17]. However, Marimbas, et al. reported that valve thrombosis is similarly frequent in patients with bio-prosthetic valves and in those with mechanical valves who are consuming adequate anticoagulant therapy [18].
Biological Aortic Valve Thrombosis
The incidence of biological prostheses in middle-aged patients has increased in the last decade due to the improved durability of newer generation prostheses, the reduced risk of reoperation for a degenerated aortic biological prosthesis, and emerging data on the unsuccessful outcome of TAVI in high-risk patients [19]. Although biological prostheses do not require anticoagulant therapy, they are subject to structural valve degeneration, resulting in limited durability, which lasts between 10-15 years and exposes the patient to the risk of re-intervention [20]. Surgical biological AV prostheses are made of biological tissue and can be xenogenic or allogenic as well as stented or stentless [21]. Metaanalysis of aortic bio-prosthesis estimates the incidence of PTV at 0.03 per 100 patients per year [22]. Due to similar transvalvular flow gradients, mechanical and transcatheter valves are homogeneous in terms of thrombogenic potential while bio-prosthesis remains the least thrombogenic prosthetic valve [23].
Transcatheter Aortic Valve Thrombosis
Since the first TAVI was performed in 2002, the rate of this procedure has risen enormously, with more than 50 000 having been performed worldwide, the vast majority of them in Europe [24]. In cases of severe aortic stenosis and appropriate heart anatomy, TAVI is the best treatment option [25]. Although current literature declares that SAVR is more likely to cause bleeding complications, acute kidney insufficiency, and new arrhythmias, TAVI is associated with higher rates of permanent pacemaker implantation, paravalvular regurgitation, and vascular complications [26,27].
During the TAVI procedure, the tissue valve is implanted by balloon dilation or self-expansion. They are inserted percutaneously, using transfemoral access in 90% of cases, and the atrioventricular angle during the procedure must be >60 degrees [28,29]. PVT after TAVI is rare, with an estimated incidence of approximately 0.61%-2.8% based on echocardiography or cardiac CT angiography [30]. Risk factors for PVT include balloon-expandable valves, large valve prosthesis size, inappropriate valve expansion, patient-prosthesis mismatch, and valve-in-valve TAVI [31]. Researchers are currently attempting to prove that stasis in the neosinus between the native AV leaflets and the TAVI leaflets may be a major contributor to the formation of PVT, and quantification of stasis using patient-specific in vitro modeling has shown that it correlates with the extent of thrombosis [32]. Bio-prosthetic valves, both surgical and transcatheter, have better hemodynamic properties than mechanical valves, therefore, antithrombotic therapy is needed during the endothelialization process in the first 3-6 months after surgery. Although oral anticoagulants have been shown to reduce the incidence of leaflet thrombosis, the routine use of an oral anticoagulant after TAVI leads to excessive bleeding, and studies are currently underway to investigate whether the use of anticoagulants therapy will be required after TAVI in the future [33].
Indications for Mitral Valve Replacement and Risk Factors
Based on 2020 ACC/AHA guidelines for the management of patients with VHD, Mitral Valve (MV) surgery is indicated for symptomatic severe primary Mitral Regurgitation (MR) when LVEF is above 30% and asymptomatic severe primary MR in patients with severe, chronic disease and left ventricular dysfunction with LVEF of 30% to 60%, or LVESD of at least 40 mm. For secondary MR valve replacement is indicated when patients do not have severe ventricular dysfunction and are symptomatic despite optimal medical therapy or while undergoing concomitant coronary artery bypass grafting [20]. Myllykangas study in Finland that included 3684 patients shows that the annual prevalence rate of mitral surgery during the study period was 3.9/100 000. 62.4% of these procedures were MV repairs and 37.6% were MV replacements. The mean age of MV surgery patients is 66.0 years and 33.4% of the patients were women [34]. The ideal time for MV surgery is when the patient‘s left ventricle approaches but has not yet reached the parameters that indicate systolic dysfunction (LVEF ≤ 60% or LVESD ≥ 40 mm) [35]. MV replacement is recommended for patients with moderate to severe mitral stenosis (mitral valve area ≤ 1.5 cm2) who have NYHA class III or IV symptoms, are not at high risk for surgery, and have an MV not amenable to either percutaneous mitral ballon commissurotomy or open commissurotomy/repair [20]. The opening and closing angles determined by 64-slice multidetector CT were 10.9 ± 0.6° and 131.1 ± 3.2° for the MV [36]. The main factors for the development of mitral PVT are hemodynamic (local blood flow turbulences, relatively slow blood flow in case of AFib, atrial dilation or low LV output), hemostatic (tissue injury), surface (leaflet injury, prosthesis malposition, incomplete prosthesis endothelialization) [37].
Mechanical Mitral Valve
Mitral PVT is 2-3 times more common than aortic prosthetic thrombosis. The annual rate of PVT ranges from 0.1%- 5.7%, with higher rates in the early perioperative period and in association with sub-therapeutic anticoagulation [38]. For patients undergoing prosthetic MV replacement surgery, the decision regarding the choice of prosthesis mechanical or bio-prosthetic is based on the need for anticoagulation with mechanical valves, the belief in improved durability with current bio-prostheses, and patient risks for reoperation [39]. Mortality rates of patients who receive a mechanical valve are equal to those of patients who receive a bio-prosthetic valve. Mechanical Prosthetic Valves (MPV) are associated with a lower risk of reoperation but an increased risk of bleeding and stroke [40]. Lifelong anticoagulation using oral Vitamin K Antagonists (VKA) is required for all currently available mechanical MV prostheses. Newer anticoagulants have not been shown to be safe or effective in patients with MPV. Anticoagulation with a VKA might be reached with acceptable risk in most patients younger than 50 years, particularly if there is appropriate monitoring of INR levels. Mechanical valve in patients <50 years of age is chosen more frequently due to a more favorable balance between valve durability and risk of bleeding and thromboembolic events, unless anticoagulation is not desired, cannot be monitored, or is contraindicated [20]. The intensity of anticoagulation depends on the characteristics of the prosthetic valve, e.g., bileaflet or tilting disc, and patient factors such as a history of thromboembolism, AFib, left atrial enlargement, left ventricular dysfunction with the target INR of 3.0 for modern bileaflet mechanical MV prostheses [41].
Biological Mitral Valve
Biological heart valves should be chosen in patients with shorter anticipated survival or comorbidities that may lead to reoperations and those who are at increased risk for bleeding. Bio-prostheses are recommended for patients >70 years of age or those in whom life expectancy is shorter than the expected bioprosthetic valve durability. Thromboembolic complications are less frequent in pregnant women with biological bio-prostheses than mechanical [42]. Butnaru‘s case reported an incidence of 6% mitral bio-prosthetic valve thrombosis out of 149 MV replacement surgeries over 10 year period, two-thirds of whom showed resolution of symptoms [43]. A significant amount of implanted mitral bio-prosthesis degenerate over time and needs further treatment due to clinically significant prosthetic stenosis or regurgitation [44]. Between 60%-70% of patients who undergo MV replacement surgery receive a bioprosthetic valve [39]. It was reported that patients between 40 to 69 years of age who receive a bio-prosthetic MV have significantly higher 15-year mortality rates compared with those who received a mechanical MV [45].
Transcatheter Mitral Valve Replacement
Together with developing transcatheter MV repair, investigators are improving methods for Transcatheter MV Replacement (TMVR) [46]. Compared with transcatheter AV replacement, TMVR has been more challenging because of the size, location, and anatomy of the MV, and the risk of left ventricular outflow tract obstruction [47]. TMVR for patients who have severe primary MR is a safe alternative when there are contraindications for surgery or operative risk is high [48]. As patients are carefully selected to avoid the failure of intervention, rejection rates for TMVR have been about 60% to 70%. Recent developments demonstrate the promise of increasing the population that could be treated, for example, using a transseptal or transapical approach. The procedure success rate was high but has some limitations due to intra-cardiac maneuverability and sheath sizes [49]. Advancing the development of TMVR is critical, as MR is the most common valvular lesion in the world, and substantial advances in the treatment of Heart Failure (HF) have led to an aging population with MR who may be at either high or prohibitive risk for open surgery [50].
Mechanical Tricuspid Valve
According to 2020 ACC/AHA guidelines, surgical treatment for Tricuspid Valve (TV) is an option for patients with Tricuspid Regurgitation (TR) at the time of surgery for left-sided valve lesions to treat symptomatic or asymptomatic severe regurgitation or to prevent further development of severe regurgitation. Surgical intervention should also be considered for selected patients with isolated TR in the absence of Pulmonary Hypertension (PH) or dilated cardiomyopathy. MPV is rarely implanted in the right heart, mainly because of its important thrombogenicity. TV replacement is needed when a repair is unfeasible because of the marked leaflet tethering, complex primary TR with tricuspid annulus diameter >40 mm, or severe tricuspid stenosis with valve area ≤ 1.0 cm2 with the manifesting symptoms of right ventricular malfunction, such as peripheral edema and ascites [51]. The mean age of the surgery is 52.3 ± 8.8 years and 66.7% are females [52]. Thrombosis is the most important and common complication of the prosthetic TV and is 20 times more likely in the tricuspid position (the reason is the lower pressure of the right system, the right ventricular morphology, and the low prostacyclin concentration of venous blood) during the first post-operative year comparing with the AV and MV [53]. Mechanical valves have desirable hemodynamic properties, low gradients, low disturbances inflow, and long durability. The normal range of the opening angle is 60°-80° for tilting disc valves and 73°-90° for bileaflet valves [54]. MPV is preferred in younger age groups as well as patients with another mechanical valve [55].
Biological Tricuspid Valve
The advantage of the bio-prosthetic valves is that they do not require long-life anticoagulation in contrast to the mechanical valves and cause a higher risk for thromboembolism and hemorrhage [56]. Because of the lower risk of thromboembolism, many surgeons favor bioprosthetic valves but the choice of the type of prosthesis should be individual, considering the patient’s age, condition, and concomitant cardiac or non-cardiac diseases [57]. Low pressure and stress in the right heart provide higher durability for the bio-prosthesis compared to the left-sided prosthetic valves and requires fewer reoperations compared with the mechanic valve as well [58]. The average time for failure of tricuspid bio-prosthesis is 7 years [59]. There is no difference between the mechanical and tissue valves in survival rate. Patients who receive bio-prostheses must be carefully evaluated with periodic echocardiograms, especially after 7 to 10 years, for detecting clinical and subclinical signs of structural valve degeneration [60].
Clinical Presentation and Diagnosis
The clinical presence of PVT varies depending on whether there is an obstruction. Severe Obstructive PVT (OPVT) is usually associated with HF, while a Non-Obstructive PVT (NOPVT) is often an accidental finding or presents as an embolic episode. When the obstruction is partial it results in dyspnea, systemic embolism, or fever. In the presence of fever diagnostic blood cultures should be performed to prevent or to early diagnose infectious endocarditis. Concentrations of D-dimers may be affected and increased. A detailed physical examination should be performed, examining for the suppression or disappearance of prosthetic sounds and the occurrence of new regurgitating or obstructive murmurs, especially if PVT is suspected for the first time. The initial diagnostic tests are Transthoracic Echocardiogram (TTE) and cine fluoroscopy [61]. Echocardiographic features that might help to suspect PVT are an acute increase in valve gradient of more than 50% over 5 years, thickening of the valve leaflet (>2 mm) downstream of the valve, decreased or limited leaflet mobility, and regress of echocardiographic abnormalities within 1-3 months of anticoagulant therapy. Cinefluoroscopy helps to assess the movement of the leaflets in mechanical valves. Nevertheless, for cine fluoroscopy, a suitable patient position is required and this examination is time-consuming. Conversely, the valve can be reoriented in any CT plane. CT offers optimal visualization of valve profiles and it does not depend on valve position or patient physical characteristics. CT complements TTE and cine fluoroscopy by performing a functional and morphological evaluation of the mechanical valve as it combines the measured values of the opening and closing angles with other information like the size of the valve or the presence of a thrombus or pannus [62].
Thrombosis of Prosthetic Heart Valves: Treatment
Main therapeutic modalities considered when PVT is diagnosed: surgery, fibrinolysis, heparin treatment, or optimization of anticoagulant and antiplatelet therapy. Treatment depends on the presence of obstruction and prosthesis location but prosthesis type does not have importance in choosing a treatment.
Right-Sided Obstructive and Non-Obstructive PVT
Right-sided thrombosis is much more frequent than the left side. Although there are no formal prospective studies evaluating different treatment modalities, intensified anticoagulation should be the first choice of treatment in patients with right-sided NOPVT. Patients with obstructive TV thrombosis usually present with signs of right HF, such as peripheral edema and ascites, and the prosthetic valve click may be inaudible during the auscultation. TTE and Transesophageal Echocardiography (TEE) can reveal the increased echogenicity on the prosthesis, decreased movement of the disc, and an elevated prosthetic valve gradient. The treatment of choice in right-sided OPVT is thrombolytic therapy, fibrinolytic agents are associated with a high success rate and a low complication rate. Surgery should be reserved for cases of thrombolytic failure, and contraindication to thrombolysis. Replacement of the mechanical TV with a bio-prosthesis can be considered in patients with failed thrombolysis, recurrent thrombosis, evidence of pannus, or contraindications to thrombolytic therapy [63].
Non-Obstructive Left-Sided PVT
Management can be divided according to the size of the thrombus. Fibrinolysis has been performed with success for small non-obstructive thrombi, but also with a significant risk of systemic embolism. The prognosis is favorable with medical therapy by optimization of anticoagulant treatment (short-term intravenous unfractionated heparin followed by warfarin adjustment and aspirin addition) for small asymptomatic thrombi (length <10 mm). If thrombus size is increased or is complicated by embolism, thrombolytic therapy or surgery should be considered [64]. In all cases of PVT, anticoagulant treatment has to be optimally adjusted. Surgery is recommended for large (≥ 10 mm) NOPVT complicated by embolism or which persists despite optimal anticoagulation [65]. Fibrinolysis should also be considered if surgery is at high risk.
Left-Sided Obstructive PVT
The treatment includes surgery (thrombectomy or valve replacement), thrombolytic therapy, and heparin. After a diagnosis has been made, heparin treatment should be started immediately. Surgery is recommended for patients in NYHA functional classes III-IV unless it is at high risk. Surgery is also indicated for critically ill patients and restricts thrombolysis to patients with high surgical risk (thrombus area (2D TEE) >0.8 cm2 and thrombus diameter 1.0 cm is associated with increased embolic risk and the rate of complications increases by 2.4 times per 1.0 cm2 increase in size, which makes surgery a better option [66]. Outcomes have been better with thrombolytic therapy; therefore, it is suggested that thrombolytic therapy would be the treatment of choice in all cases except for patients with contraindications to these agents.
In hemodynamically unstable patients fibrinolysis should be consisting of recombinant tissue Plasminogen Activator (rtPA) 10 mg bolus +90 mg in 90 mins, or streptokinase 1 500 000 U in 60 mins without heparin. While in hemodynamically stable patients: urokinase 4500 U/kg/h over a 12 h period, or 2000 U/kg/h+heparin over 24 h, streptokinase 500 000 IU in 20 mins followed by 1 500 000 IU for 10 h without heparin or rtPA 10 mg bolus, 50 mg during the first hour, 20 mg during the second hour and 20 mg during the third hour [61].
Prevention
Long-term anticoagulation is required to prevent thromboembolism after mechanical heart valve replacement, its value in patients receiving bio-prostheses is uncertain [37]. Moderate-intensity anticoagulant therapy (INR of 1.5 to 2.0) was reported to be safe and feasible for preventing thromboembolic complications in high-risk surgical patients who are receiving long-term oral anticoagulant therapy. The study included 18% of patients with a mechanical valve [67]. Patients with mechanical or bio-prosthetic valves should receive antibiotic prophylaxis before some dental and surgical procedures to prevent endocarditis [68]. Nowadays, a new generation of smart heart devices is capable of auto-detecting valve status and able to measure specific markers in plasma that could predict prosthetic HF. For bioprostheses, several markers have been identified as predictors of structural valve degeneration: the ratio apolipoprotein B and A-I (apoB/apoA-I); lipoprotein-associated phospholipase A2 (Lp-PLA); the ratio of oxidized low-density lipoprotein and high-density lipoprotein (OxLDL/HDL); proprotein convertase subtilisin/kexin 9 (PCSK9) levels [69]. In addition, to solve geometry issues like patient-prosthesis mismatch latest technologies use stereolithographic 3D printing models based on X-ray CT scans of native valves [70]. Using this technology it becomes possible to produce a tailor-made prosthetic valve, made of tissues or polymers that would mimic the native valve with a minimal error. In patients with suspected bioprosthetic valve thrombosis who are hemodynamically stable, a trial of anticoagulation should be considered. It is also crucial to recognize the difference between structural valve degeneration and bioprosthetic valve thrombosis as a cause of prosthetic valve dysfunction as the treatment differ. Thrombolysis or secondary surgical intervention could be suggested in patients who do not respond to anticoagulation therapy or are hemodynamically unstable [71].
Case Report
Case 1
A 48 years old woman was referred to Lithuanian University of Health Sciences (LUHS) Kaunas Clinics, Department of Cardiology suffering shortness of breath during minimal exertion, significantly reduced exercise tolerance, swelling of the legs, and recurrence of rhythm disorders. The patient has been suffering from the above complaints for the past year, with shortness of breath and HF symptoms progressing during the last months. In 1999, the patient was diagnosed with a mixed rheumatic defect and underwent mechanical AV and MV prosthesis, since then she has been using warfarin in therapeutic norms. The patient had recurrent Atrial Flutter (AF) episodes since 2016 and had undergone radiofrequency catheter ablation 4 times but no positive effect was observed. VVI mode cardiac pacing was implanted in 2019, followed by recurrence of atypical AF episodes which was reversed with Cordarone. During TTE severe TR was observed. For further investigation into the function of the mechanical prostheses, treatment of TR, and clarification of the diagnosis, a decision to hospitalize the patient was obtained. The patient denied other chronic and infectious diseases. For outpatient treatment, she has been using warfarin, Cordarone, β-blockers, trimetazidine, and diuretics.
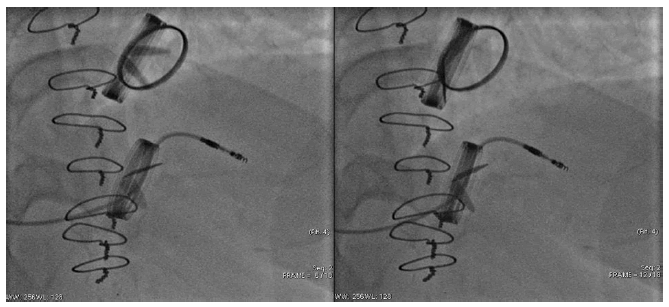
Upon arrival in the cardiology department, clinical findings included: Heart Rate (HR) of 83 bpm., Blood Pressure (BP) of 120/70 mmHg, pulse oximetry of 96%, normal mechanical valve clicks, and an II° systolic murmur at the apex of the heart, AV and Erb’s point. Laboratory tests showed slightly elevated NT-proBNP (344.9 ng/l) and anticoagulation studies revealed an INR of 3.1, an aPTT of 45.1s, and a PT of 80.6s. ECG showed normal cardiac pacing activity. 2D TTE showed impaired LVEF-50%, elevated transprosthetic aortic Doppler peak velocity-3.2 m/s, elevated peak transprosthetic gradient-40.96 mmHg, I° intraprosthesis regurgitation. Signs of rheumatic damage to the TV were also observed, such as copulating leaflets, accelerated tricuspid peak velocity-1.7 m/s, III-IV° TR. Chest x-ray showed mild pulmonary vascular congestion. Invasive hemodynamic tests were performed to differentiate possible causes of dyspnoea and severe post-capillary PH with reduced cardiac output was found. TTE was repeated to monitor the dynamics-AV prosthesis peak velocity (3.8-4) m/s was recorded from the adaptive positions and assessed as prosthesis dysfunction (Figure 1).
The patient’s status worsened and the “heart team” Concilium recommended reoperation of the AV and TV prosthesis surgery. In the department of cardiology lansoprazole and heparin, with dose titration according to aPPT, were added to the previous treatment. The patient was transferred to the cardiac surgery unit for further surgical treatment-aortic root and TV prosthesis surgery in case of dysfunction of the mechanical AV prosthesis and severe TV regurgitation (Figure 2).
PVT can still occur despite adequate anticoagulation and optimal INR values, so it is important to monitor patients at risk and identify complications as soon as possible.
Case 2
A 60-year-old man was referred to the LUHS Kaunas Clinics, Department of Cardiology with progressive dyspnea on minimal exertion and at rest, cough that produces hemoptysis and weakness. The previous medical history of the patient was a surgical closure of patent ductus arteriosus at the age of 8 and rheumatism in childhood. There is a family history of rheumatism. A mechanical AV for severe aortic stenosis was implanted 15 years ago and the patient was treated with acenocoumarol since. Complaints lasted for a month; especially it has been intensifying during the last 2 weeks. In the regional hospital, the patient was treated for organizing pneumonia and progressing respiratory failure which was recognized as complications of the COVID-19 infection. He was treated with dexamethasone and cefuroxime but it was unsuccessful. There is a history of left middle cerebral artery infarct that was diagnosed in 2016, the patient lost eyesight in one eye since. The patient was admitted to taking beta-blockers, Angiotensin Receptor- Neprilysin inhibitors (ARNi), and loop diuretics due to HF, hypertension, and permanent AFib. Hospitalization was required for the further investigation of progressing respiratory and HF.
Upon arrival in the cardiology department, clinical findings included: HR 94 b.p.m., BP 116/54 mmHg, pulse oximetry 92% with 6 L/min oxygen mask, irregular heartbeats, both diastolic and systolic III° murmurs at the projection of the aortic, tricuspid valves, apex and Erb’s point.
Laboratory results showed electrolytes in normal range, normal liver function and impaired renal function (creatinine 150.8 mcmol/l, urea 20.7 mmol/l). Troponin I was 82 ng/L (n<14 ng/L), NT-proBNP 18000 ng/L (n<300 ng/L), D-dimers 2400 mcg/L (n<350 mcg/L). Abnormal coagulation test was observed with INR 2.7, PT 67.1 s, aPTT 41s. ECG-AFib.
2D TTE showed reduced mobility of the one-leaflet mechanical aortic prosthesis with an image suggestive of thrombus (V max-4,6 m/s, G max-73,96 m/s, the mean trans-prosthetic gradient-29 mmHg, effective orifice area-0.97 cm2) and reduced LVEF (44%) in the setting of AFib. Dilated left ventricle, moderate (II°-III°) MV insufficiency, signs of severe PH-right ventricle systolic pressure-89 mmHg.
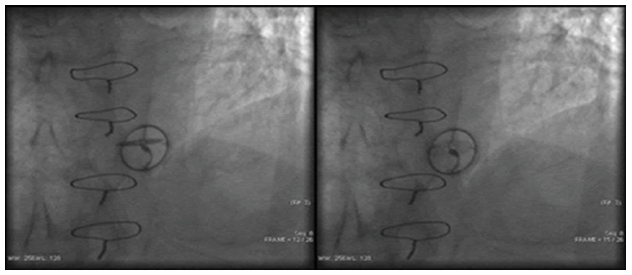
Chest X-ray showed large bilateral non-homogenous infiltrations. The AV opening angle measured by the thoracic CT scan was 15° (Figure 3). Coronary artery angiography was performed to differentiate the cause of dyspnea but coronary artery stenoses were excluded and it confirmed the dysfunction of AV.
Despite the adequate treatment of HF, and anticoagulation therapy, the patient’s condition was worsening due to venous congestion and hypotension, therefore urgent surgical treatment was recommended. Intravenous dopamine was given to the patient but the cardiogenic shock was progressing and the patient was transferred to the intensive care unit. The mechanical aortic prosthesis was surgically removed and changed with a bio-prosthetic valve. During the postoperative period, the patient’s condition continued to worsen due to progressing respiratory failure, cardiogenic shock, acute renal failure, liver failure, urinary tract infection, and metabolic acidosis. Patient vital functions were supported with venoarterial extracorporeal membrane oxygenation, renal replacement therapy, and mechanical ventilation; antibiotics were added to the treatment. On the 8th postoperative day, the patient died. Histopathological examination of AV showed a thrombotic mass with the features of degeneration on the valve. Even though further investigations are needed because of the unclear mechanisms but it is suspected that the pro-inflammatory and procoagulant states of the SARS-CoV-2 virus are related to the risk of PVT despite therapeutic INR and optimal treatment.
Discussion
Prosthetic valve obstruction by pannus, thrombus, or both is one of the most life-threatening complications of mechanical prostheses [72]. Clinical presentation of the patient may vary from mild dyspnea to severe respiratory failure and rapid worsening of HF. Sudden increases in transvalvular gradients from baseline during TTE are indicative of valvular obstruction [73]. It is crucial to be aware of a possible acute decline in clinical status and alert in case of an emergency intervention. Fibrinolytic therapy and surgical intervention remain the main options in PVT management, even though it is still controversial and the choice is based on the clinical manifestations. The choice of the PVT treatment is reliant on the features of mechanical valve thrombosis [9]. High-risk left-sided PVT features (NYHA functional class III to IV, mobility of the thrombus, and the size >0.8 cm2) are in favor of the surgical approach.
Thrombosis may present with acute manifestations while the pannus usually presents with chronic evolution, over several weeks or months. Prosthetic valve dysfunction at the AV position is often caused by pannus formation which is an uncommon, but serious complication. Its incidence varies between 1.8% in tilting discs to 0.73% in bileaflet valves [74]. All types of available prosthetic valves can be affected by pannus formation. Pannus formation is a biological reaction to valve prostheses associated with factors such as surgical technique, and thrombus formation due to inadequate anticoagulation and infection [72].
Rheumatic heart disease patients usually are treated with mechanical valve replacement surgery, owing to their durability and lower reoperation rates. However, these patients require lifelong oral anticoagulation therapy to prevent thromboembolic complications. Chronic anticoagulation has the risk of bleeding which necessitates close monitoring of INR. This complicates the lives of patients with a mechanical prosthesis, thus inadequate anticoagulation therapy is the main reason for the development of PVT. A large meta-analysis of 1005 patients showed that up to 61% of patients with PVT had adequate anticoagulation so the cause of PVT in them remains unclear [75]. However, the INR can be falsely elevated in certain cases. Falsely elevated INR has been recorded in patients with lupus anticoagulant antibodies or dysfibrinogenemia and those undergoing conversion from direct thrombin inhibitors [76]. Though the increase in INR may also result from coagulation factors deficiency, liver dysfunction, or vitamin K deficiency [77]. In our first case, the patient had sufficient anticoagulation but still developed prosthetic valve dysfunction. In this case, we need to remain alert and suspect other conditions that may be responsible for the high INR values.
Cardiac rheumatism may have played a role in our cases, especially considering that our first patient also developed TV rheumatic regurgitation. It is important to note that most of the previously reported cases of pannus formation at the AV position were females as well as our first case. Differential diagnosis between thrombus and pannus is made based on clinical, echocardiographic, and morphopathological criteria and is essential because therapeutic approaches are different. If in thrombotic obstruction, thrombolysis can be considered, this is contraindicated in the case of pannus, where the only valid treatment is the surgery-valve replacement or pannus removal [78]. In our case, chronic evolution and effective anticoagulation resemble the clinical picture of pannus formation rather than thrombosis. Since a TV prosthesis surgery was required, it was decided that the dysfunction of the AV prosthesis would be corrected during the same surgery.
Recently it was discovered that the pro-coagulant effect of the SARS-CoV-2 virus does not always protect patients from the risk of thrombosis, even under effective anticoagulation like was observed in a second clinical case. In patients with mechanical valves receiving VKA treatment, a high risk of thrombus formation remains [79]. The treatment is based on thrombolysis by therapeutic anticoagulation, fibrinolysis, or surgery and is selected depending on the size, composition of thrombus, and clinical status. In our case, due to the manifestation of organizing pneumonia as a latephase COVID-19 infection complication and the rapidly worsening condition of the patient, surgical treatment was required. Allione’s study shows that the monitoring of D-dimer levels associated with the inflammation parameters (CRP and IL-6) might be useful for early recognition of prothrombotic patterns associated with SARS-CoV-2, despite therapeutic values of INR during warfarin treatment [80].
Conclusion
Despite PVT being rare, it is a highly lethal condition that requires rapid diagnostic assessment. Diagnosis is based on the findings of the clinical examination, cine fluoroscopy, and echocardiography. The role of TTE is fundamental, not only for accurate diagnosis but also to provide optimal treatment. Treatment depends on the presence of obstruction and prosthesis location and might be chosen as one of the following: surgery, fibrinolysis, heparin treatment, or optimization of anticoagulant and antiplatelet therapy. Adequate anticoagulation remains the most important factor in the prevention of PVT in the mechanical prosthesis.
Declarations
Conflict of Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
- Capodanno, Davide, et al. "Standardized definitions of structural deterioration and valve failure in assessing long-term durability of transcatheter and surgical aortic bioprosthetic valves: A consensus statement from the European Association of Percutaneous Cardiovascular Interventions (EAPCI) endorsed by the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS)." European Heart Journal, Vol. 38, No. 45, 2017, pp. 3382-90.
Google Scholar Crossref - Yaminisharif, Ahmad, Mohammad Javad Alemzadeh-Ansari, and Seyed Hossein Ahmadi. "Prosthetic tricuspid valve thrombosis: Three case reports and literature review." The Journal of Tehran University Heart Center, Vol. 7, No. 4, 2012, pp. 147-55.
Google Scholar Crossref - Kalpana, S. R., et al. "Prosthetic valve thrombosis-Association of genetic polymorphisms of VKORC1, CYP2C9 and CYP4F2 genes." Medicine, Vol. 98, No. 6, 2019, p. e14365.
Google Scholar Crossref - Lim, Wei Yao, Guy Lloyd, and Sanjeev Bhattacharyya. "Mechanical and surgical bioprosthetic valve thrombosis." Heart, Vol. 103, No. 24, 2017, pp. 1934-41.
Google Scholar Crossref - Sondergaard, Lars, et al. "The year in cardiology 2017: Valvular heart disease." European Heart Journal, Vol. 39, No. 8, 2018, pp. 650-57.
Google Scholar Crossref - Lung, Bernard, et al. "A prospective survey of patients with valvular heart disease in Europe: The Euro heart survey on valvular heart disease." European Heart Journal, Vol. 24, No. 13, 2003, pp. 1231-43.
Google Scholar Crossref - Lawrie, Gerald M. "Role of Transcatheter Aortic Valve Implantation (TAVI) versus conventional aortic valve replacement in the treatment of aortic valve disease." Methodist DeBakey Cardiovascular Journal, Vol. 8, No. 2, 2012, pp. 4-8.
Google Scholar Crossref - Yacoub, M. H., and J. J. M. Takkenberg. "Will heart valve tissue engineering change the world?" Nature Clinical Practice Cardiovascular Medicine, Vol. 2, No. 2, 2005, pp. 60-61.
Google Scholar Crossref - Vahanian, Alec, et al. "2021 ESC/EACTS Guidelines for the management of valvular heart disease: developed by the Task Force for the management of valvular heart disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS)." European Heart Journal, Vol. 43, No. 7, 2022, pp. 561-632.
Google Scholar Crossref - Musumeci, Lucia, et al. "Prosthetic aortic valves: Challenges and solutions." Frontiers in Cardiovascular Medicine, Vol. 5, 2018, p. 46.
Google Scholar Crossref - Chakravarty, Tarun, et al. "Subclinical leaflet thrombosis in surgical and transcatheter bioprosthetic aortic valves: An observational study." The Lancet, Vol. 389, No. 10087, 2017, pp. 2383-92.
Google Scholar Crossref - Martinod, Kimberly, and Denisa D. Wagner. "Thrombosis: Tangled up in NETs." Blood, The Journal of the American Society of Hematology, Vol. 123, No. 18, 2014, pp. 2768-76.
Google Scholar Crossref - Zaidi, Mariam, et al. "Aortic valve surgery: Management and outcomes in the paediatric population." European Journal of Pediatrics, Vol. 180, No. 10, 2021, pp. 3129-39.
Google Scholar Crossref - Pibarot, Philippe, and Jean G. Dumesnil. "Prosthetic heart valves: Selection of the optimal prosthesis and long-term management." Circulation, Vol. 119, No. 7, 2009, pp. 1034-48.
Google Scholar Crossref - Stein, Paul D., et al. "Antithrombotic therapy in patients with mechanical and biological prosthetic heart valves." Chest, Vol. 119, No. 1, 2001, pp. 220S-27.
Google Scholar Crossref - Patel, Akash, and Sagar Patel. "To lyse or not to lyse: Mechanical aortic valve thrombosis." Chest, Vol. 158, No. 4, 2020, p. A188.
Google Scholar Crossref - Ma, Wei-Guo, et al. "Dysfunction of mechanical heart valve prosthesis: Experience with surgical management in 48 patients." Journal of Thoracic Disease, Vol. 7, No. 12, 2015, pp. 2321-29.
Google Scholar Crossref - Maribas, Philippe. "Management of prosthetic heart valve obstruction: Speech for the surgery?" Archives of Cardiovascular Diseases, Vol. 102, No. 4, 2009, pp. 255-57.
Google Scholar Crossref - Kaneko, Tsuyoshi, Lawrence H. Cohn, and Sary F. Aranki. "Tissue valve is the preferred option for patients aged 60 and older." Circulation, Vol. 128, No. 12, 2013, pp. 1365-71.
Google Scholar Crossref - Otto, Catherine M et al. “2020 ACC/AHA guideline for the management of patients with valvular heart disease: Executive summary: A report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines.” Circulation, Vol. 143, No. 5, 2021, pp. e35-71.
Google Scholar Crossref - Head, Stuart J., Mevlut Celik, and A. Pieter Kappetein. "Mechanical versus bioprosthetic aortic valve replacement." European Heart Journal, Vol. 38, No. 28, 2017, pp. 2183-91.
Google Scholar Crossref - Puvimanasinghe, J. P. A., et al. "Prognosis after aortic valve replacement with a bioprosthesis: Predictions based on meta-analysis and microsimulation." Circulation, Vol. 103, No. 11, 2001, pp. 1535-41.
Google Scholar Crossref - Scotten, Lawrence N., and Rolland Siegel. "Thrombogenic potential of transcatheter aortic valve implantation with trivial paravalvular leakage." Annals of Translational Medicine, Vol. 2, No. 5, 2014.
Google Scholar Crossref - Clayton, Benjamin, G. Morgan-Hughes, and C. Roobottom. "Transcatheter Aortic Valve Insertion (TAVI): A review." The British Journal of Radiology, Vol. 87, No. 1033, 2014, p. 20130595.
Google Scholar Crossref - Cahill, T. J., et al. "Transcatheter aortic valve implantation: Current status and future perspectives." European Heart Journal, Vol. 39, No. 28, 2018, pp. 2625-34.
Google Scholar Crossref - Leon, Martin B., et al. "Transcatheter or surgical aortic-valve replacement in intermediate-risk patients." New England Journal of Medicine, Vol. 374, No. 17, 2016, pp. 1609-20.
Google Scholar - Arora, Sameer, Jacob A. Misenheimer, and Radhakrishnan Ramaraj. "Transcatheter aortic valve replacement: Comprehensive review and present status." Texas Heart Institute Journal, Vol. 44, No. 1, 2017, pp. 29-38.
Google Scholar Crossref - Tchetche, Didier, et al. "How to make the TAVI pathway more efficient." Interventional Cardiology Review, Vol. 14, No. 1, 2019, pp. 31-33.
Google Scholar Crossref - Naganuma, Toru, et al. "Successful use of the loop snare technique for crossing a degenerated surgical valve with the Evolut‐R system." Catheterization and Cardiovascular Interventions, Vol. 93, No. 7, 2019, pp. E400-02.
Google Scholar Crossref - Jose, John, et al. "Clinical bioprosthetic heart valve thrombosis after transcatheter aortic valve replacement: Incidence, characteristics, and treatment outcomes." JACC: Cardiovascular Interventions, Vol. 10, No. 7, 2017, pp. 686-97.
Google Scholar Crossref - Khodaee, Farhan, et al. "Incomplete expansion of transcatheter aortic valves is associated with propensity for valve thrombosis." Interactive CardioVascular and Thoracic Surgery, Vol. 30, No. 1, 2020, pp. 39-46.
Google Scholar Crossref - Trusty, Phillip M., et al. "The role of flow stasis in transcatheter aortic valve leaflet thrombosis." The Journal of Thoracic and Cardiovascular Surgery, 2020.
Google Scholar Crossref - Rogers, Toby, et al. "Randomized trial of aspirin versus warfarin after transcatheter aortic valve replacement in low-risk patients." Circulation: Cardiovascular Interventions, Vol. 14, No. 1, 2021, p. e009983.
Google Scholar Crossref - Myllykangas, Monna E., et al. "Population trends in mitral valve surgery in Finland between 1997 and 2014: The finnish CVD register." Scandinavian Cardiovascular Journal, Vol. 52, No. 1, 2018, pp. 51-57.
Google Scholar Crossref - Kang, Duk-Hyun, et al. "Comparison of early surgery versus conventional treatment in asymptomatic severe mitral regurgitation." Circulation, Vol. 119, No. 6, 2009, pp. 797-804.
Google Scholar Crossref - Lee, Dong-Hyeon, et al. "The measurement of opening angle and orifice area of a bileaflet mechanical valve using multidetector computed tomography." Korean Circulation Journal, Vol. 39, No. 4, 2009, pp. 157-62.
Google Scholar Crossref - Dangas, George D., et al. "Prosthetic heart valve thrombosis." Journal of the American College of Cardiology, Vol. 68, No. 24, 2016, pp. 2670-89.
Google Scholar Crossref - Lin, Steve S., et al. "Prediction of thrombus-related mechanical prosthetic valve dysfunction using transesophageal echocardiography." The American Journal of Cardiology, Vol. 86, No. 10, 2000, pp. 1097-101.
Google Scholar Crossref - Gammie, James S., et al. "Isolated mitral valve surgery: The society of thoracic surgeons adult cardiac surgery database analysis." The Annals of Thoracic Surgery, Vol. 106, No. 3, 2018, pp. 716-27.
Google Scholar Crossref - Chikwe, Joanna, et al. "Survival and outcomes following bioprosthetic vs. mechanical mitral valve replacement in patients aged 50 to 69 years." JAMA, Vol. 313, No. 14, 2015, pp. 1435-42.
Google Scholar - Lung, Bernard, and Josep Rodes-Cabau. "The optimal management of anti-thrombotic therapy after valve replacement: Certainties and uncertainties." European Heart Journal, Vol. 35, No. 42, 2014, pp. 2942-49.
Google Scholar Crossref - Jeejeebhoy, Farida Mary. "Prosthetic heart valves and management during pregnancy." Canadian Family Physician, Vol. 55, No. 2, 2009, pp. 155-57.
Google Scholar - Butnaru, Adi, et al. "Diagnosis and treatment of early bioprosthetic malfunction in the mitral valve position due to thrombus formation." The American Journal of Cardiology, Vol. 112, No. 9, 2013, pp. 1439-44.
Google Scholar Crossref - Ruel, Marc, et al. "Late incidence and determinants of reoperation in patients with prosthetic heart valves." European Journal of Cardio-Thoracic Surgery, Vol. 25, No. 3, 2004, pp. 364-70.
Google Scholar Crossref - Goldstone, Andrew B., et al. "Mechanical or biologic prostheses for aortic-valve and mitral-valve replacement." New England Journal of Medicine, Vol. 377, No. 19, 2017, pp. 1847-57.
Google Scholar - Webb, John G., et al. "Percutaneous transcatheter mitral valve replacement: First-in-human experience with a new transseptal system." Journal of the American College of Cardiology, Vol. 73, No. 11, 2019, pp. 1239-46.
Google Scholar Crossref - De Backer, Ole, et al. "Percutaneous transcatheter mitral valve replacement: An overview of devices in preclinical and early clinical evaluation." Circulation: Cardiovascular Interventions, Vol. 7, No. 3, 2014, pp. 400-09.
Google Scholar Crossref - Feldman, Ted, et al. "Percutaneous repair or surgery for mitral regurgitation." New England Journal of Medicine, Vol. 364, No. 15, 2011, pp. 1395-406.
Google Scholar - Sorajja, Paul, et al. "Initial feasibility study of a new transcatheter mitral prosthesis: The first 100 patients." Journal of the American College of Cardiology, Vol. 73, No. 11, 2019, pp. 1250-60.
Google Scholar Crossref - Fiorilli, Paul N., Howard C. Herrmann, and Wilson Y. Szeto. "Transcatheter mitral valve replacement: Latest advances and future directions." Annals of Cardiothoracic Surgery, Vol. 10, No. 1, 2021, pp. 85-95.
Google Scholar Crossref - Rodes-Cabau, Josep, Maurizio Taramasso, and Patrick T O'Gara. "Diagnosis and treatment of tricuspid valve disease: Current and future perspectives." The Lancet, Vol. 388, No. 10058, 2016, pp. 2431-42.
Google Scholar Crossref - Altaani, Haitham Akram, and Saed Jaber. "Tricuspid valve replacement, mechnical vs. biological valve, which is better?" International Cardiovascular Research Journal, Vol. 7, No. 2, 2013, pp. 71-74.
Google Scholar - Piper, C., D. Hering, and D. Horstkotte. "Prosthetic valve thrombosis: Predisposition and diagnosis." European Heart Journal Supplements, Vol. 3, No. suppl_Q, 2001, pp. Q16-21.
Google Scholar Crossref - Lancellotti, Patrizio, et al. "Recommendations for the imaging assessment of prosthetic heart valves: A report from the European Association of Cardiovascular Imaging endorsed by the Chinese Society of Echocardiography, the Inter-American Society of Echocardiography, and the Brazilian Department of Cardiovascular Imaging." European Heart Journal-Cardiovascular Imaging, Vol. 17, No. 6, 2016, pp. 589-90.
Google Scholar Crossref - Rizzoli, Giulio, et al. "Biological or mechanical prostheses in tricuspid position? A meta-analysis of intra-institutional results." The Annals of Thoracic Surgery, Vol. 77, No. 5, 2004, pp. 1607-14.
Google Scholar Crossref - Filsoufi, Farzan, et al. "Long-term outcomes of tricuspid valve replacement in the current era." The Annals of Thoracic Surgery, Vol. 80, No. 3, 2005, pp. 845-50.
Google Scholar Crossref - Shinn, Sung Ho, and Hartzell V. Schaff. "Evidence-based surgical management of acquired tricuspid valve disease." Nature Reviews Cardiology, Vol. 10, No. 4, 2013, pp. 190-203.
Google Scholar Crossref - Carrier, Michel, et al. "Tricuspid valve replacement: An analysis of 25 years of experience at a single center." The Annals of Thoracic Surgery, Vol. 75, No. 1, 2003, pp. 47-50.
Google Scholar Crossref - Kaplan, Mehmet, et al. "Prosthetic replacement of tricuspid valve: Bioprosthetic or mechanical." The Annals of Thoracic Surgery, Vol. 73, No. 2, 2002, pp. 467-73.
Google Scholar Crossref - Garatti, Andrea, et al. "Twenty-five year outcomes of tricuspid valve replacement comparing mechanical and biologic prostheses." The Annals of Thoracic Surgery, Vol. 93, No. 4, 2012, pp. 1146-53.
Google Scholar Crossref - Roudaut, Raymond, Karim Serri, and Stephane Lafitte. "Thrombosis of prosthetic heart valves: Diagnosis and therapeutic considerations." Heart, Vol. 93, No. 1, 2007, pp. 137-42.
Google Scholar Crossref - Suh, Young Joo, et al. "Measurement of opening and closing angles of aortic valve prostheses in vivo using dual-source computed tomography: Comparison with those of manufacturers' in 10 different types." Korean Journal of Radiology, Vol. 16, No. 5, 2015, pp. 1012-23.
Google Scholar Crossref - Duran, Nilufer Eksi, Murat Biteker, and Mehmet Ozkan. "Treatment alternatives in mechanical valve thrombosis." Archive of the Turkish Society of Cardiology: It is the publication of the Turkish Society of Cardiology, Vol. 36, No. 6, 2008, pp. 420-25.
Google Scholar - Kalcik, Macit, et al. "Thrombus attached to suture materials successfully thrombolysed with low-dose tissue plasminogen activator." Archives of the Turkish Society of Cardiology, Vol. 42, No. 1, 2014, pp. 61-63.
Google Scholar - Authors/Task Force Members, et al. "Guidelines on the management of valvular heart disease (version 2012) The joint task force on the management of valvular heart disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS)." European Heart Journal, Vol. 33, No. 19, 2012, pp. 2451-96.
Google Scholar Crossref - Tong, Ann T., et al. "Transesophageal echocardiography improves risk assessment of thrombolysis of prosthetic valve thrombosis: Results of the international PRO-TEE registry." Journal of the American College of Cardiology, Vol. 43, No. 1, 2004, pp. 77-84.
Google Scholar - Larson, Bradley JG, Marc S. Zumberg, and Craig S. Kitchens. "A feasibility study of continuing dose-reduced warfarin for invasive procedures in patients with high thromboembolic risk." Chest, Vol. 127, No. 3, 2005, pp. 922-27.
Google Scholar Crossref - Trujillo, John F., and Steven M. Hollenberg. "Valvular heart disease in adults: Management of prosthetic heart valves." FP Essentials, Vol. 457, 2017, pp. 23-29.
Google Scholar - Nsaibia, Mohamed Jalloul, et al. "Association between plasma lipoprotein levels and bioprosthetic valve structural degeneration." Heart, Vol. 102, No. 23, 2016, pp. 1915-21.
Google Scholar Crossref - Hockaday, L. A., et al. "Rapid 3D printing of anatomically accurate and mechanically heterogeneous aortic valve hydrogel scaffolds." Biofabrication, Vol. 4, No. 3, 2012, p. 035005.
Google Scholar Crossref - Balakrishnan, Kuhendra, Brian Herman, and George Koshy. "Very late bioprosthetic aortic valve thrombosis." BMJ Case Reports CP, Vol. 12, No. 5, 2019, p. e228871.
Google Scholar Crossref - Barbetseas, John, et al. "Differentiating thrombus from pannus formation in obstructed mechanical prosthetic valves: An evaluation of clinical, transthoracic and transesophageal echocardiographic parameters." Journal of the American College of Cardiology, Vol. 32, No. 5, 1998, pp. 1410-17.
Google Scholar - Salamon, Jason, et al. "Mechanical valve obstruction: Review of diagnostic and treatment strategies." World Journal of Cardiology, Vol. 7, No. 12, 2015, p. 875.
Google Scholar Crossref - Sakamoto, Yoshimasa, et al. "Prevalence of pannus formation after aortic valve replacement: Clinical aspects and surgical management." Journal of Artificial Organs, Vol. 9, No. 3, 2006, pp. 199-202.
Google Scholar Crossref - Zoghbi, William A., et al. "Recommendations for evaluation of prosthetic valves with echocardiography and doppler ultrasound: A report From the American Society of Echocardiography's Guidelines and Standards Committee and the Task Force on Prosthetic Valves, developed in conjunction with the American College of Cardiology Cardiovascular Imaging Committee, Cardiac Imaging Committee of the American Heart Association, the European Association of Echocardiography, a registered branch of the European Society of Cardiology, the Japanese ...." Journal of the American Society of Echocardiography, Vol. 22, No. 9, 2009, pp. 975-1014.
Google Scholar Crossref - Teshima, Hideki, et al. "Obstruction of St. Jude medical valves in the aortic position: Plasma transforming growth factor type beta 1 in patients with pannus overgrowth." Artificial Organs, Vol. 34, No. 3, 2010, pp. 210-15.
Google Scholar Crossref - Huang, Grace, et al. "Treatment of obstructive thrombosed prosthetic heart valve." Journal of the American College of Cardiology, Vol. 62, No. 19, 2013, pp. 1731-36.
Google Scholar Crossref - Cardoso, Goncalo, et al. "A rare case of two mechanisms of prosthetic valve dysfunction in the same patient." Portuguese Journal of Cardiology (English Edition), Vol. 32, No. 12, 2013, pp. 1037-41.
Google Scholar Crossref - Jeckelmann, Clarisse, et al. "Case report: Mechanical mitral prosthetic valve thrombosis in the context of COVID-19 despite effective anticoagulation." European Heart Journal-Case Reports, Vol. 6, No. 2, 2022.
Google Scholar Crossref - Allione, Attilio, et al. "Coronavirus disease 2019 (COVID-19) and prosthetic heart valve: An additional coagulative challenge." World Journal of Emergency Medicine, Vol. 11, No. 4, 2020, p. 258.
Google Scholar Crossref