Research - International Journal of Medical Research & Health Sciences ( 2021) Volume 10, Issue 9
Protective Effect of Ajwa Fruit Extract on Nicotine Induced Oviduct Morphometric Changes in Adult Albino Rats
Faeza Rauf1*, Shagufta Suhail2, Tayyaba Muzaffar3, Sadia Iqbal4, Zafar Iqbal5 and Muhammad Suhail32Department of Anatomy, Punjab Postgraduate Medical Institution, Lahore, Pakistan
3Department of Anatomy, Shaikh Zayed Postgraduate Medical Institute, Lahore, Pakistan
4Department of Anatomy, Allama Iqbal Medical College, Lahore, Pakistan
5Department of Anatomy, Rai Medical College, Sargodha, Pakistan
Faeza Rauf, Department of Anatomy, Azra Naheed Medical College, Lahore, Pakistan, Email: rauf.faeza@gmail.com
Received: 18-Aug-2021 Accepted Date: Sep 23, 2021 ; Published: 30-Sep-2021
Abstract
Nicotine alters oviduct histology and Ajwa date (Phoenix dactylifera) contains antioxidant that prevents nicotine-induced toxicity in the oviduct of adult albino rats. Objectives: To evaluate the protective effect of Ajwa date against oviduct toxicity induced by nicotine in adult albino rats. Material and Methods: This experimental study was conducted in the Department of Anatomy, Shaikh Zayed Postgraduate Medical Institute, Lahore. Adult female albino rats of average weight 250 g were randomly divided into three groups A (control group), B, and C (experimental groups), each containing 10 female albino rats. 1 ml/kg distilled water was given to the rats of the control (A) group by gastric intubation, experimental group B was given nicotine injection intraperitoneally (0.1 mg/kg body weight) for 28 days. The rats of experimental group C were given 0.1 mg/kg body weight nicotine intraperitoneally plus 1000 mg/kg body weight Ajwa fruit extract by gastric intubation for 28 days. Rats were sacrificed on the 30th day of the experiment. Fallopian tubes were carefully dissected out and fixed in 10% formalin for routine histological examination. Result: Mean oviduct wall thickness of ampulla and isthmus of nicotine treated group B (58 ± 12.5 μm, 102.4 ± 19.9 μm respectively) was significantly lower (p<0.002, p<0.001 respectively) as compared to those of control group A (84 ± 15.4 μm, 133.9 ± 9.6 μm respectively) and those of nicotine plus Ajwa date treated group C (75.9 ± 16.9 μm, 121.4 ± 12 μm respectively) while no significant difference was observed between group A and C. Mean epithelial height of ampulla and isthmus of nicotine treated group B (15.4 ± 3.5 μm, 16.3 ± 3.1 μm respectively) was significantly lower as compared to the height of epithelium in control group A (23.1 ± 1.49 μm, 24.4 ± 3. 8 μm, respectively) and nicotine plus Ajwa treated group C (20.8 ± 3.4 μm, 20.7 ± 2.5 μm, respectively). The difference in the mean of epithelial height between three groups of ampulla and isthmus was significant (p<0.001*). The number of mucosal folds in ampulla and isthmus of group B (8.3 ± 2.8, 5.6 ± 1.8, respectively) was much less than group A (15.0 ± 3.6, 8.7 ± 2.4) and group C (13.7 ± 3.7, 7.1 ± 1.6). The difference in the mean of the number of mucosal folds among the three groups was significant (p<0.001*). Conclusion: Nicotine-induced toxic effect on the oviduct can be reduced by an antioxidant activity following administration of Ajwa date.
Keywords
Nicotine, Epithelial height, Oviduct, Phoenix dactylifera
Introduction
The trend of Shisha use and smoking goes on increasing in the youth of our country. A study from a local university narrates that 43% male and 11% female students were found to be a regular users of shisha [1]. An ordinary cigarette contains 7 mg to 22 mg of nicotine, out of which 1 mg is absorbed by the smoker. An average hookah/shisha bowl contains nicotine equivalent to that of 20 cigarettes [2].
Nicotine is also available in other forms like gum, sublingual tablets, oral capsules, oral solution, nasal spray, inhaler, transdermal patch, intravenous and subcutaneous injections, and enema. After inhalation, nicotine is rapidly dispersed in the bloodstream and reaches the blood-brain barrier within 10-20 seconds, and damages the central nervous system [3-6]. Nicotine has a half-life of 6-8 hours and it gathers in blood during the day and reduces during sleep. The lethal dose orally for an adult human is (30-60) mg of nicotine. In children, LD50 is around 10 mg [7]. For rats, LD50 of nicotine is 50 mg/kg and for mice is 3 mg/kg [8].
Nicotine affects germ cell structure and function in males as well as in females. It causes degeneration of seminiferous tubules, alters spermatogenesis, and reduces testosterone level by reducing Steroidogenic Acute Regulatory Protein (StAR), the protein necessary for testosterone biosynthesis [9-11]. In the case of nicotine-treated female rats, there is a reduction in estrogen-dependent parameters including uterine weight, endometrial and myometrial thickness, and diameter. Nicotine causes luteal insufficiency by inhibiting progesterone release. Nicotine inhibits gonadotropin release from the pituitary. It also alters the concentration of steroid hormone and causes irregularities in the estrous cycle. Nicotine impairs ovulation, decreases the number and size of ovarian follicles and corpora lutea. The number of atrophic follicles also rises [12].
In one study about Rhesus monkeys, oviductal blood flow was reduced by nicotine due to decreased smooth muscle contraction in the wall of the oviduct, interrupting the transport of the embryo [13]. Structural and functional damage of the oviduct can cause tubal pregnancy or infertility [14,15]. About 78% of ectopic pregnancies occur in the ampulla of the fallopian tube [16]. Orally administered nicotine affected oocyte growth, fertilization, and early pregnancy in mice [17].
Palms “the princess of the plant kingdom,” is one of the main fruit crops in the Arab countries, the Middle East, and Africa. Dates are a staple food, a main source of income and play an important part in their civilization, and economy. Several benefits of Ajwa dates are known to us [18,19]. Arabs called it a tree of life and is a symbol of richness and affection to Muslims [20]. Ajwa fruit is the fleshy pericarp, rich in sugars 65%-80%, is a highly nutritious food. Date fruit has protein without starch and is a rich source of fibers, low in fat, minerals, and vitamins. Proteins are 1%-3% with their favorable amino acid pattern for human needs. Date consumption is high among Muslims, especially in Arabic countries. Date Palm Pollen (DPP) is a fountain of youth and has been used as a rejuvenating medicinal agent in early Egyptians and Chinese history. DPP increases dietary intake and acts as a diet supplement. Date pits gave in animal feed to improve their growth. For dietary purposes, pollen preparations are distributed worldwide. Date seeds are potential sources of pharmaceuticals and edible oils [21].
Date pit extract reduces women’s wrinkles [22]. P. dactylifera L is useful in treating cough, bronchitis, rheumatism, burning sensation, nephropathy, and sexual incapacity [23]. In a study, DPP raised serum values of estrogen and progesterone. Estrogenic activity is due to the flavonoid and alkaloid parts of DPP. Ovarian diameter, the number of ovarian follicles (primary, secondary and Graafian), and corpora lutea were increased after DPP administration in female mice. DPP suspension increases oogenesis in the embryo when given during pregnancy and lactation [24].
The increasing trend of smoking among female youth is causing issues related to fertility. Raised levels of nicotine alter oviduct histology. Ajwa dates (Phoenix dactylifera) contain an antioxidant so may prevent nicotine toxicity. That’s why this study was designed to evaluate the protective effect of Ajwa date against oviduct toxicity induced by nicotine in the isthmus and ampulla regions of the oviduct.
Materials and Methods
After the approval of the institutional animal ethical committee, this study was conducted in the Department of Anatomy, Shaikh Zayed Postgraduate Medical Institute, Lahore in collaboration with the Department of Anatomy, Punjab PGMI, Lahore. 30 adult healthy female albino rats aged 3-4 months (average body weight 200 g-250 g) were kept at (23 ± 2)ºC and a 12 h light and the dark cycle were maintained. They were fed on normal food and given water ad libitum. They were allowed to acclimatize for two weeks before the experiment started. Ajwa dates were purchased from Madinah Al-Munawara, and their extract was prepared by the following method (Ali A and Abdu S) [25].
3000 gram of date fruits were manually separated from pits and 1-liter distilled water was added to this coarsely pounded date fruit (3:1). This was left for 48 h in a refrigerator (4°C) with occasional stirring. This aqueous extract of Ajwa dates contains phenolic content (1.752 mg gallic Acid equivalent/g of extract) and flavonoid content (0.1239 mg quercetin equivalent/g of extract) quantified by the Chemistry Department, PCSIR, Lahore. Nicotine synthesis grade (99% pure) loose liquid Fluka, was purchased from Sigma-Aldrich chemical company (USA).
The animals were randomly divided into three groups by lottery method, each containing ten animals. Rats of control group A were treated with 1 mL/kg body-weight of distilled water by gastric intubation for 28 days. To experimental group B, nicotine was given 0.1 mg/kg body-weight/day intraperitoneally for 28 days. In experimental group C, each rat was given nicotine 0.1 mg/kg body-weight/day intraperitoneally and Ajwa fruit extract 1000 mg/kg body-weight by gastric intubation for 28 days.
48 hours after giving the last dose, rats were humanly sacrificed by using intraperitoneal administration of morphine at a dose of (0.3-0.5) mg/kg. The estrous cycle was determined by the vaginal smear method and only those rats which were in an estrous phase were dissected, other rats were left till their estrous phase. Both fallopian tubes were removed for investigation, kept in 10% neutral buffered formaldehyde solution for 48 hours. Small pieces were cut, and paraffin blocks were made. Serial sections of 5 μm thickness were taken. Hematoxylin and Eosin-stained oviduct were observed under the light microscope.
Quantitative variables include wall thickness, the height of epithelium, and the number of mucosal folds in the ampulla and isthmus of fallopian tubes. The thickness of the wall was measured from basement membrane to serosa in three different fields and their mean was calculated. The height of the oviduct epithelium was taken from the upper limit of the cell facing the lumen to the basement membrane in three different fields and their mean was calculated. The number of the mucosal folds was counted in the ampulla and isthmus.
Statistical Analysis
Data was entered and evaluated by using Statistical Package for Social Sciences (SPSS), version 20. Wall thickness, the height of the epithelium, and the number of mucosal folds in the ampulla and isthmus were described as mean ± S.D. The normality of the data was assessed by the Shapiro Wilk test. As data was normally distributed therefore one way ANOVA test was used to compare the wall thickness, height the of epithelium, and number of mucosal folds in ampulla and isthmus among groups. p-value ≤ 0.05 was considered significant.
Results
Tissue sections from the ampulla and isthmus of the fallopian tubes in the estrus stage were selected for the study. All animals of healthy control group A showed normal mucosa with multiple folds each lined by ciliated columnar and non-ciliated secretory cells resting on a basement membrane. The oviduct wall consisted of three layers including inner mucosa, middle muscularis layer, and outer serosa.
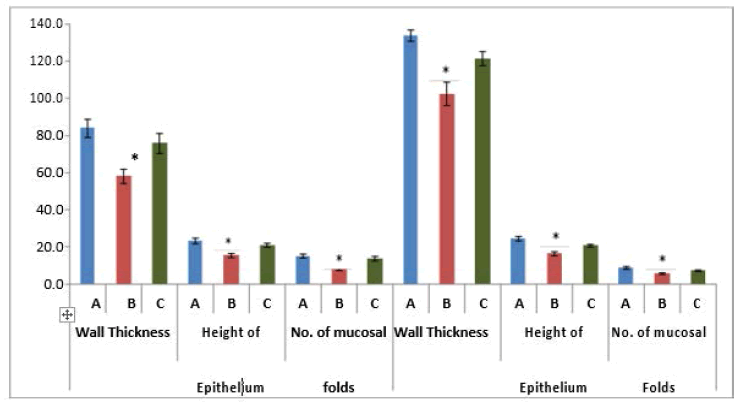
The mean oviduct wall thickness of the ampulla of control group A was calculated as 84 ± 15.4 μm. The mean oviduct wall thickness of the ampulla of experimental group B was calculated as 58 ± 12.5 μm and of group C was 75.9 ± 16.9 μm. Results revealed that the mean wall thickness of ampulla of group B was significantly lower as compared to groups A and C while no significant difference was observed between groups A and C. Difference in the mean of three groups of the oviduct wall thickness of ampulla was significant (p<0.002*) (Figure 1).
The mean oviduct wall thickness of isthmus of control group A was 133.9 ± 9.6 μm while of experimental group B was 102.4 ± 19.9 μm and of the group, C was 121.4 ± 12 μm. Results revealed that the mean wall thickness of the Isthmus of group B was significantly lower as compared to group A and C while no significant difference was observed between group A and C. While Mean wall thickness of ampulla was quite lower than that of the isthmus of group B. Difference in the mean of three groups of the oviduct wall thickness of isthmus was significant (p<0.001*) (Figure 1).
The epithelial height of the ampulla of the fallopian tube of control group A was calculated as 23.1 ± 1.49 μm. The mean epithelial height of ampulla of the fallopian tube of group B was 15.4 ± 3.5 μm and of the group, C was 20.8 ± 3.4 μm respectively (Figure 1).
Height of the epithelium in the isthmus of the fallopian tube of control group A was 24.4 ± 3.8 μm and for group B and C, it was 16.3 ± 3.1 μm and 20.7 ± 2.5 μm The results revealed that mean epithelial height of ampulla and Isthmus of group B was significantly lower as compared to the height of epithelium in group A and C while no significant difference was observed between group A and C. In the ampulla of group B, epithelial height was lower than in the isthmus (Figure 1).
The difference in the mean of epithelial height among three groups of ampulla and isthmus was significant (p<0.001*) (Figure 1).
The number of mucosal folds in the ampulla of group A was calculated as 15.0 ± 3.6, while in groups B and C were 8.3 ± 2.8 and 13.7 ± 3.7. In group B, mucosal folds were much less than the other two groups. The difference in the mean number of mucosal folds among three groups of ampulla was significant (p<0.001*) (Figure 1).
In an isthmus of group A, the number of mucosal folds was calculated as 8.7 ± 2.4, while in groups B and C were 5.6 ± 1.8 and 7.1 ± 1.6. In group B, mucosal folds were less in number than the other two groups. The difference in the mean number of mucosal folds among three groups of the isthmus was significant (p<0.006*) (Figure 1).
Discussion
Healthy endosalpinx and a balanced local hormone environment are essential for the functions of the oviduct. The fimbriated infundibulum captures and forces the ovulated oocytes into the oviduct. In the ampulla, fertilization and early embryonic cleavage occur. The isthmus regulates the transport of sperm to the ampulla as well as the passage of early embryos to the uterus.
In the given study, rats were sacrificed in the estrous phase, in which maturation of ovarian follicles occurs due to a high level of estrogen. Ciliary movement and stability of oviduct epithelium are regulated by changes in estradiol and progesterone levels [26,27]. In epithelial cells, gonadotropins regulate progesterone receptors expression in nuclei of non-ciliated cells and at the base of cilia [28-30]. Ciliated-estrogen receptor protein is universally expressed in all regions of a tube in rats [31].
Estrogen regulates ciliary motility, enhances ciliation and secretion in the oviduct [32]. Progesterone is associated with deciliation, atrophy, and muscle contraction [33]. In the control group, due to the high estrogen level in the estrous phase, epithelial height was more in isthmus than in ampulla as compared to experimental groups B and C.
Nicotine is the present study caused a reduction in height of epithelial cells of ampulla and isthmus of the oviduct in group B, as compared to the control group A and Ajwa date extract plus nicotine group C. Nicotine prevents steroidogenesis [34]. Patra, et al. stated that nicotine will either hinder the capability of estrogen to control follicular development or manufacture estrogen [35]. Iranloye and Bolarinwa reported that nicotine increased the length of the estrous cycle with the introduction of a new phase “subestrous”, which hindered ovulation in women smokers [36]. Nicotine causes a hypoestrogenic state by inhibiting 21 hydroxylases [37]. Nicotine decreased estrogen levels and led to reduced cellular height.
Decreased epithelial height can change the function of the oviduct. Normally fertilization occurs in the ampulla of the fallopian tube. In nicotine treated group, epithelial height was less in the ampulla than that in the isthmus, it can lead to ectopic pregnancy or infertility in smokers. These results coincide with results of research by Patil, et al., Tuttle, et al. and Rauf F, where the nicotine administration reduced the number and size of the secondary follicle, Graafian follicles, and corpora lutea and increased the number of atrophic follicles [14,38,39].
In control group A, the number of mucosal folds and mean wall thickness was more than those of groups B and C. These results coincide with a study showing increased height and number of primary folds and epithelial thickness in the infundibulum and ampulla during the follicular phase [40].
The number of mucosal folds, wall thickness, and epithelial height in isthmus and ampulla of fallopian tubes was comparatively reduced in experimental group B than that in the control and Ajwa date extract plus nicotine treated group. The reduction in the oviduct reserves can be explained by the fact that nicotine activated the nicotinic acetylcholine receptors, to increase Ca2+ ion influx, which in turn increased the formation of Reactive Oxygen Species (ROS) causing oxidative stress in the cell [41]. ROS caused DNA damage and led to apoptosis [42,43].
P. dactylifera L exerts its protective effect by reducing malondialdehyde, a marker for oxidative stress [44]. P. dactylifera L enhances the actions of antioxidant enzymes such as glutathione reductase, glutathione S-transferase, catalase, glutathione peroxidase, and superoxide dismutase [45]. Literature has shown the presence of powerful non-enzymatic antioxidants, phenolics, flavonoid compounds, vitamins A, C, and E in Ajwa dates [46]. Both powerful non-enzymatic and enzymatic antioxidants detoxified ROS and chelated different radicals through enzymatic and non-enzymatic reactions to reduce DNA destruction and oxidative stress thus ameliorating the toxic effects of nicotine on oviduct in group C. Similar results are seen in the study by Rauf F, where Ajwa fruit extract reduced the histopathological lesions remarkably [39]. The result also coincides with the results of the Abdu SB study where the Ajwa dates showed protective influence against ochratoxin A induced testicular damage [47]. Keeping in view all these benefits, Ajwa date fruit can be labeled as medicinal food [48].
Conclusion
The current research work concludes that the toxic effect of nicotine was reduced by the antioxidant activity of Ajwa date extract. Hence, it can be recommended as a protective agent against nicotine-induced oviduct toxicity due to its easy, rapid, and safe dietary administration.
Declarations
Conflicts of Interest
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Declarations
Conflicts of Interest
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
References
- Khan, Javaid. "Tobacco epidemic in Pakistan." Journal of Postgraduate Medical Institute (Peshawar-Pakistan), Vol. 26, No. 3, 2012.
- Aslam, Hafiz Muhammad, et al. "Harmful effects of shisha: Literature review." International Archives of Medicine, Vol. 7, No. 1, 2014, pp. 1-9.
- Agarwal, Ashok, and Tamer M. Said. "Oxidative stress, DNA damage and apoptosis in male infertility: A clinical approach." BJU International, Vol. 95, No. 4, 2005, pp. 503-07.
- Andersson, Christer, Paula Wennström, and Jorn Gry. "Nicotine alkaloids in Solanaceous food plants." Nordic Council of Ministers, 2003.
- Balfour, D., et al. "Diagnosis and treatment of nicotine dependence with emphasis on nicotine replacement therapy. A status report." European Heart Journal, Vol. 21, No. 6, 2000, pp. 438-45.
- Tyndale, Rachel F., and Edward M. Sellers. "Variable CYP2A6-mediated nicotine metabolism alters smoking behavior and risk." Drug Metabolism and Disposition, Vol. 29, No. 4, 2001, pp. 548-52.
- Schep, Leo J., Robin J. Slaughter, and D. Michael G. Beasley. "Nicotinic plant poisoning." Clinical Toxicology, Vol. 47, No. 8, 2009, pp. 771-81.
- Okamoto, Masato, et al. "Effects of aging on acute toxicity of nicotine in rats." Pharmacology & Toxicology, Vol. 75, No. 1, 1994, pp. 1-6.
- Jana, Kuladip, Prabhat Kumar Samanta, and Dipak Kumar De. "Nicotine diminishes testicular gametogenesis, steroidogenesis, and steroidogenic acute regulatory protein expression in adult albino rats: Possible influence on pituitary gonadotropins and alteration of testicular antioxidant status." Toxicological Sciences, Vol. 116, No. 2, 2010, pp. 647-59.
- Mendelson, Jack H., et al. "Effects of intravenous cocaine and cigarette smoking on luteinizing hormone, testosterone, and prolactin in men." Journal of Pharmacology and Experimental Therapeutics, Vol. 307, No. 1, 2003, pp. 339-48.
- Oyeyipo, Ibukun P., Yinusa Raji, and Adeyombo F. Bolarinwa. "Nicotine alters male reproductive hormones in male albino rats: The role of cessation." Journal of Human Reproductive Sciences, Vol. 6, No. 1, 2013, pp. 40-44.
- Tuttle, Anne Mulligan, Martin Stämpfli, and Warren G. Foster. "Cigarette smoke causes follicle loss in mice ovaries at concentrations representative of human exposure." Human Reproduction, Vol. 24, No. 6, 2009, pp. 1452-59.
- Wong, Michael K., et al. "Adverse effects of perinatal nicotine exposure on reproductive outcomes." Reproduction, Vol. 150, No. 6, 2015, pp. R185-93.
- Sadan, Oscar, et al. "Role of steroid receptors in the pathogenesis of tubal pregnancy." The Journal of Reproductive Medicine, Vol. 47, No. 12, 2002, pp. 1031-34.
- Ajonuma, Louis Chukwuemeka, et al. "Ultrastructural characterization of whole hydrosalpinx from infertile Chinese women." Cell Biology International, Vol. 29, No. 10, 2005, pp. 849-56.
- Land, Jolande A., and Jan W. Arends. "Immunohistochemical analysis of estrogen and progesterone receptors in fallopian tubes during ectopic pregnancy." Fertility and Sterility, Vol. 58, No. 2, 1992, pp. 335-37.
- Jin, Z., et al. "Effects of nicotine administration on elemental concentrations in mouse granulosa cells, maturing oocytes and oviduct epithelium studied by X-ray microanalysis." Journal of Submicroscopic Cytology and Pathology, Vol. 30, No. 4, 1998, pp. 517-20.
- Ragab, Ahmed R., et al. "Antioxidant and tissue-protective studies on Ajwa extract: Dates from al-Madinah al-Monwarah, Saudia Arabia." Journal of Environmental and Analytical Toxicology, Vol. 3, No. 1, 2013.
- Al-Qarawi, Aly Abdullah, et al. "Protective effect of extracts from dates (Phoenix dactylifera L.) on carbon tetrachloride-induced hepatotoxicity in rats." International Journal of Applied Research in Veterinary Medicine, Vol. 2, No. 3, 2004, pp. 176-80.
- AbdAlla, M. M., and Aesha M. Abd El-Kawy. "Karyotype analysis for date palm (Phoenix dactylifera L) compared with tissue culture derived plants." New York Science Journal, Vol. 3, No. 11, 2010, pp. 165-70.
- Biglari, Foroogh, Abbas FM AlKarkhi, and Azhar Mat Easa. "Antioxidant activity and phenolic content of various date palm (Phoenix dactylifera) fruits from Iran." Food Chemistry, Vol. 107, No. 4, 2008, pp. 1636-41.
- Bauza, E., et al. "Date palm kernel extract exhibits antiaging properties and significantly reduces skin wrinkles." International Journal of Tissue Reactions, Vol. 24, No. 4, 2002, pp. 131-36.
- Selvam, A. "Inventory of vegetable crude drug samples housed in botanical survey of India, Howrah." Pharmacognosy Reviews, Vol. 2, No. 3, 2008, pp. 61-94.
- Moshfegh, Fazeleh, et al. "Effects of date palm pollen on fertility and development of reproductive system in female Balb/C mice." Journal of Herbmed Pharmacology, Vol. 5, No. 1, 2015, pp. 23-28.
- Ali, Awatef, and Susan Abdu. "Antioxidant protection against pathological mycotoxins alterations on proximal tubules in rat kidney." Functional Foods in Health and Disease, Vol. 1, No. 4, 2011, pp. 118-34.
- Hill M.A. "Embryology Estrous Cycle." 2021. https://embryology.med.unsw.edu.au/embryology/index.php/Estrous_Cycle
- Quarmby, Lynne M. "Cellular deflagellation." International Review of Cytology, Vol. 233, 2004, pp. 47-92.
- Hunter, R. H. F., B. Flechon, and J. E. Flechon. "Distribution, morphology and epithelial interactions of bovine spermatozoa in the oviduct before and after ovulation: A scanning electron microscope study." Tissue and Cell, Vol. 23, No. 5, 1991, pp. 641-56.
- Teilmann, Stefan Cuoni, et al. "Expression and localization of the progesterone receptor in mouse and human reproductive organs." Journal of Endocrinology, Vol. 191, No. 3, 2006, pp. 525-35.
- Wessel, Th, U. Schuchter, and H. Walt. "Ciliary motility in bovine oviducts for sensing rapid non-genomic reactions upon exposure to progesterone." Hormone and Metabolic Research, Vol. 36, No. 03, 2004, pp. 136-41.
- Shao, Ruijin, et al. "Ciliated epithelial-specific and regional-specific expression and regulation of the estrogen receptor-β2 in the fallopian tubes of immature rats: A possible mechanism for estrogen-mediated transport process in vivo." American Journal of Physiology-Endocrinology and Metabolism, Vol. 293, No. 1, 2007, pp. E147-58.
- Reeder, Richard L., and Barbara Shirley. "Deciliation in the ampulla of the rat oviduct and effects of estrogen on the process." Journal of Experimental Zoology, Vol. 283, No. 1, 1999, pp. 71-80.
- Akison, L. K., and R. L. Robker. "The critical roles of Progesterone Receptor (PGR) in ovulation, oocyte developmental competence and oviductal transport in mammalian reproduction." Reproduction in Domestic Animals, Vol. 47, 2012, pp. 288-96.
- Sanders, Sara R., S. Peder Cuneo, and Adele M. Turzillo. "Effects of nicotine and cotinine on bovine theca interna and granulosa cells." Reproductive Toxicology, Vol. 16, No. 6, 2002, pp. 795-800.
- Patra, P. B., S. Sanyal, and N. M. Biswas. "Possible alpha-adrenergic involvement in nicotine induced alteration of spermatogenesis in rat." Andrologia, Vol. 11, No. 4, 1979, pp. 273-78.
- Iranloye, B. O., and A. F. Bolarinwa. "Effect of nicotine administration on estrous cycle in female albino rats." Nigerian Journal of Health and Biomedical Sciences, Vol. 6, No. 2, 2007, pp. 21-25.
- Jin, Z., and G. M. Roomans. "Effects of nicotine on the uterine epithelium studied by X-ray microanalysis." Journal of Submicroscopic Cytology and Pathology, Vol. 29, No. 2, 1997, pp. 179-86.
- Patil, Somanathreddy, et al. "Effect of graded doses of nicotine on ovarian and uterine activities in albino rats." Indian Journal of Experimental Biology, Vol. 37, No. 2, 1999, pp. 184-86.
- Rauf, Faeza, et al. "Ajwa improves nicotine induced ovarian toxicity in murine model." Proceedings, Vol. 34, No. 4, 2020, pp. 21-29.
- Mokhtar, Doaa M. "Microscopic and histochemical characterization of the bovine uterine tube during the follicular and luteal phases of estrous cycle." Journal of Microscopy and Ultrastructure, Vol. 3, No. 1, 2015, pp. 44-52.
- Hogg, R. C., Mario Raggenbass, and Daniel Bertrand. "Nicotinic acetylcholine receptors: From structure to brain function." Reviews of Physiology, Biochemistry and Pharmacology, Vol. 147, 2003, pp. 1-46.
- Sudheer, Adluri Ram, et al. "Protective effect of ferulic acid on nicotine-induced DNA damage and cellular changes in cultured rat peripheral blood lymphocytes: A comparison with N-acetylcysteine." Toxicology in vitro, Vol. 21, No. 4, 2007, pp. 576-85.
- Cheng, Shun-Feng, et al. "Nicotine exposure impairs germ cell development in human fetal ovaries cultured in vitro." Aging (Albany NY), Vol. 10, No. 7, 2018, pp. 1556-74.
- Al Harthi, S. S., et al. "Quantification of phenolic compounds, evaluation of physicochemical properties and antioxidant activity of four date (Phoenix dactylifera L.) varieties of Oman." Journal of Taibah University Medical Sciences, Vol. 10, No. 3, 2015, pp. 346-52.
- El-Far, A. H., et al. "Date palm (Phoenix dactylifera): Protection and remedy food." Current Trends in Nutraceuticals, Vol. 1, No. 2, 2016, pp. 1-10.
- Hoseinifar, Seyed Hossein, et al. "Effect of dietary supplementation of palm fruit extracts on the transcriptomes of growth, antioxidant enzyme and immune‐related genes in common carp (Cyprinus carpio) fingerlings." Aquaculture Research, Vol. 48, No. 7, 2017, pp. 3684-92.
- Abdu, Suzan Bakr. "Ameliorative influence of Ajwa dates on ochratoxin A-induced testis toxicity." Journal of Microscopy and Ultrastructure, Vol. 6, No. 3, 2018, pp. 134-38.
- Vayalil, Praveen K. "Date fruits (Phoenix dactylifera Linn): An emerging medicinal food." Critical Reviews in Food Science and Nutrition, Vol. 52, No. 3, 2012, pp. 249-71.