Research - International Journal of Medical Research & Health Sciences ( 2021) Volume 10, Issue 6
Role of CaMKII in Controlling the Morphology and Motility of Neurite Growth Cone in Lymnaea stagnalis Neurons
Atiq Hassan1, Nazim Nasir1*, Khursheed Muzammil2, Mesheil Alalyani3 and Mohammed Rehan Asad42Department of Public Health, College of Applied Medical Sciences, Khamis Mushait Campus, King Khalid University (KKU), Abha, Kingdom of Saudi Arabia
3College of Applied Medical Sciences, Al-Ghad University, Abha, Kingdom of Saudi Arabia
4Department of Basic Sciences, College of Medicine, Majmaah University, Kingdom of Saudi Arabia
Nazim Nasir, Department of Basic Medical Sciences, College of Applied Medical Sciences, Khamis Mushait Campus, King Khalid University (KKU), Abha, Kingdom of Saudi Arabia, Email: drnnasir@gmail.com
Received: 13-May-2021 Accepted Date: Jun 22, 2021 ; Published: 30-Jun-2021
Abstract
Calcium ion (Ca2+) and Calmodulin (CaM) are important signaling molecules that have been shown to play a significant role in a wide variety of neuronal functions, including neurite outgrowth of axon and dendrites. The growth cone is located on the tip of the growing neurite. The morphology and motility of the growth cone depend on Ca2+ and the stimulation of Ca2+ dependent protein kinases (CaMK) in developing neurons. CaM-kinase II (CaMKII) is a member of the CaMK family, and it is highly expressed in the cytosol of developing neurons, including the growth cone. We hypothesized that CaMKII activity could be necessary for growth cones morphology and motility. To test this possibility, the identified Pedal A (PeA) neurons from the central ring ganglia of the freshwater pond snail Lymnaea stagnalis were cultured for 24 hrs -48 hrs in a 2 ml brain conditioned medium. Following neurite outgrowth, we have examined the effect of CaMKII inhibitor on growth cone structure and motility rate by using a CaMKII specific inhibitor KN-93 and its inactive analog KN-92; both drugs were pressure applied directly onto individual growth cones. Here we demonstrate that inhibition of CaMKII dramatically changes the morphology and motility of growth cone in cultured neurons. Growth cones of neurons treated with KN-93 exhibit collapsed morphology. In contrast, growth cone exposure to KN-92 did not affect growth cone morphology and motility rate. Taken together, the data presented here provide the first direct evidence for the involvement of CaMKII activity to maintain growth cone morphology and motility in the freshwater pond snail Lymnaea stagnalis.
Keywords
Calcium, Cytoskeleton, Kinase, Lymnaea stagnalis, Neurons, Neurite growth cone
Introduction
Axonal degeneration is commonly observed following nervous system injury and other neurodegenerative diseases like Alzheimer’s Disease (AD) brain and spinal cord injury. Traumatic nervous system injuries initiate several cellular and molecular changes in and around the injury site. These cellular and molecular changes contribute to axonal damage and neuronal and non-neuronal cell death, leading to functional deficits [1,2]. Degeneration of axons occurs both below and above the level of injury [1]. Many therapies and strategies have been used to enhance and promote axonal regeneration. Regenerative therapies focus on promoting axonal regrowth and neuronal replacement through the application of growth and neurotrophic factors, applying chemicals and neutralizing the inhibitory molecules to make growth permissive environment, cell transplantation, and application of various paradigms of electrical stimulation [3-12].
Electrical Stimulation (ES) plays a vital role in a variety of common and severe health problems [4]. ES has become one of the most promising therapies for various neurological disorders [3,13,14]. ES such as epidural electrical stimulation, transcranial electrical stimulation, and direct current stimulation has successfully demonstrated the neuronal axon’s elongation and regeneration [3,15-18]. ES also induces axonal outgrowth toward the cathode, with axons aligning with the direction of current flow. ES can alter the neuronal cell membrane potential and causes the depolarization of the cell membrane which ultimately assembles the actin filaments in the growth cones cytoskeleton to promote neurite growth [19,20]. In addition, ES changes the electric fields of extracellular matrix proteins which eventually promote elongation of neurite and growth cones [19,21].
Despite the significant progress in neuroscience and regenerative medicine, however, effective nerve repair and regeneration of axons to restore normal function following nerve injury remain a challenge to the research scientist. In the last few decades, tissue engineering introduced the use of natural and synthetic biomaterials or nanomaterials to create a favorable microenvironment to promote the regeneration of nerve and axon [6,22]. Among these biomaterials, conductive materials have been used to mediate the stimulation of electrical currents to regulate cell and tissue growth [20,23]. A variety of conductive materials, including Graphene Oxide (GO), alone or in combinations have been extensively used in nerve regeneration to promote axon and neurite growth [20,23,24].
Currently, promising bio-applications of graphene-based biomaterial in neural network regeneration and nervous system repairing have drawn much attention because the unique electrical properties of graphene can effectively contribute to the electrical stimulations of neurons and the regeneration of nervous systems [20,25]. The previous study has shown that graphene and graphene oxide can alter neuronal cell behaviors, including attachment of growth cone, growth, proliferation, and differentiation of the neuronal cell [25,26]. Graphene can increase the local concentration of extracellular matrices such as collagen, laminin, and fibronectin. Recently, nanocarbon materials such as graphene and carbon nanotubes have been considered new effective electrode materials with high conductivity [13,20]. A recent study has shown that graphene oxide conductive materials combined with electrical stimulation enhance axonal extension and growth [20,25,27].
Numerous studies have demonstrated that graphene-based nano-materials play a significant role in promoting neuronal axon regeneration, neurite outgrowth, and differentiation of various cells, including neuronal stem cells into neuronal cells [20,24,28,29]. Another research has shown that graphene-based nano-materials efficiently enhanced the expression of Growth-Associate Protein-43 (GAP-43), promoting neurite sprouting and outgrowth of the growth cone to the maximal extent in growing hippocampal cultures without compromising the morphology and viability of cells [22,28].
Axonal regeneration is the primary failure to regain function following axonal injury and neurodegenerative diseases; therefore, it is critically needed to understand the cellular mechanism of neurite outgrowth and the morphology and motility of the growth cone. The previous study has shown that calcium influx occurred following axonal injury and this influx of extracellular calcium plays a vital role in the growth of axons [30]. The Calcium ions (Ca2+) play a significant role in signal transduction pathways that control various neuronal functions such as neurite outgrowth of the axon, dendrites, and synaptic plasticity [31]. Ca2+ signaling is mediated through several Ca2+-binding proteins, including Calmodulin (CaM). CaM is a regulatory protein that modulates the activity of several signaling molecules such as Protein Kinase C (PKC), phosphorylase kinase, myosin light chain kinase, calcineurin, and family of Ca2+/calmodulindependent protein kinase (CaMK) [32]. The previous study has demonstrated that overexpression of CaMKII promotes neurite outgrowth of neuroblastoma cells [33].
Multifunctional, Ca2+/calmodulin-dependent protein kinase or (CaM kinase II) is one of the calmodulin-regulated enzymes that mediates its effect through calcium signaling in neurons [34-36]. CaMKII is one of the most highly expressed proteins in neurons, comprising 1%-2% of the total protein concentration [37]. The unique properties of CaMKII, including its abundance, multifunctional nature, key location, and sophisticated regulation, may allow the enzyme to take part in important synaptic functions, including neurotransmitter synthesis and release, modulation of ion channels activity, synaptic plasticity, and gene expression [38-40].
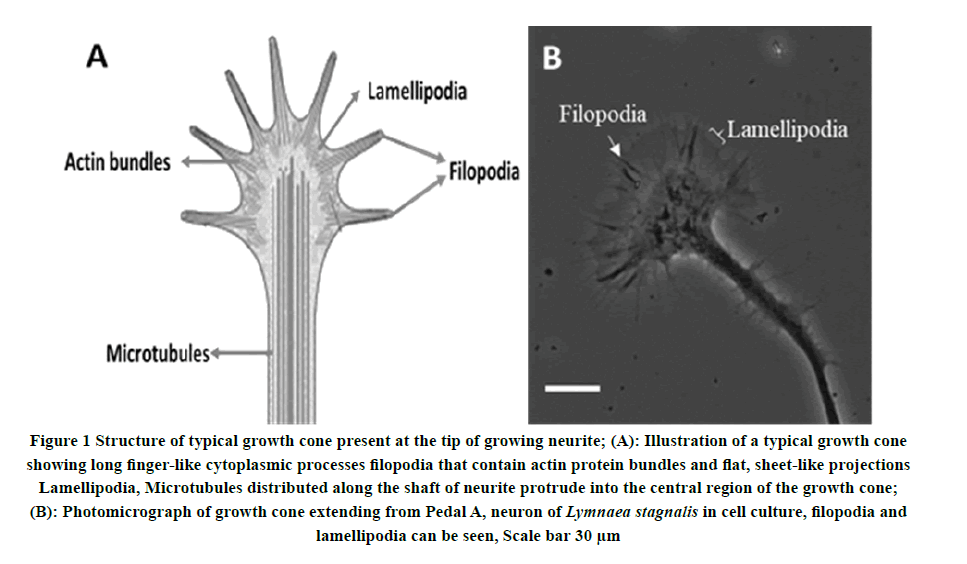
Growth cones are the motile, well-organized dynamic structure located on the motile tips of growing axons and neurites. Growth cones play a critical role in axonal pathfinding to navigates and directs axonal elongation to find out their appropriate synaptic connections during neural development [41,42]. The growth cone is a conical-shaped structure comprising finger-like projection called filopodia containing actin protein and a membranous structure known as lamellipodia (Figure 1). Filopodia and lamellipodia, both structures are necessary for the motility and advancement of the growth cone [34,43]. Growth cones establish the direction of axonal extension by detecting and responding to complex guidance cues and signaling molecules such as cell surface and extracellular matrix molecules including Netrins, Slits, Semaphorins, and Ephrins in the nervous system environment [44,45]. Growth cones are involved in neurite or axonal pathway finding in the developing nervous system [46-49]. Research in the past several decades has identified several families of chemical ligands and their corresponding receptors on growth cone and downstream signaling cascades involved in neuronal outgrowth and guidance during neuronal development [48]. These environmental cues can be chemoattractive or chemorepulsive and can steer extending neuronal growth cones at both long and short ranges, during neural development [50]. Previous research has shown that chemical ligands activate the receptors on growth cones that ultimately initiate the intracellular signals cascade to direct cytoskeletal changes [51].
Figure 1. Structure of typical growth cone present at the tip of growing neurite; (A): Illustration of a typical growth cone showing long finger-like cytoplasmic processes filopodia that contain actin protein bundles and flat, sheet-like projections Lamellipodia, Microtubules distributed along the shaft of neurite protrude into the central region of the growth cone; (B): Photomicrograph of growth cone extending from Pedal A, neuron of Lymnaea stagnalis in cell culture, filopodia and lamellipodia can be seen, Scale bar 30 μm
The cytoskeleton of the growth cone comprises Actin and tubulin microtubule; this cytoskeleton of the growth cone is highly sensitive to change in the extracellular environment [52]. The changes in the organization of the Actin and tubulin in the cytoskeleton cause the change in morphology and motility of the growth cone [53-55]. The behavior, morphology, and motility of the growth cone are highly sensitive to changes in calcium concentration in the growth cone. The morphology and motility of the growth cone depend on Ca2+ and the stimulation of Ca2+ dependent protein kinases (CaMK) in developing neurons [34,43,56]. CaMKII is highly expressed in the cytosol of developing neurons, including the growth cone. Therefore, we hypothesize that CaMKII activity could regulate axonal regeneration and maintain the growth cones morphology and motility. In this study, we have investigated the role of multifunctional key synaptic signaling protein CaMKII in maintaining and controlling the morphology and motility of growth cone in Pedal A neurons of freshwater snail Lymnaea stagnalis.
Taken together, the present research provided first direct evidence that CaMKII activity is necessary to maintain growth cone morphology and the motility rate of the growth cone of growing neurite of identified neurons of freshwater snail Lymnaea stagnalis. Moreover, it enhances our knowledge to understand the mechanisms of neurodegenerative diseases like Alzheimer’s Disease (AD) and following axonal injury.
Materials and Methods
Animals
The freshwater snail, Lymnaea stagnalis were maintained at room temperature in well-aerated, de-chlorinated tap water, and kept at a 12/12 hours light/dark cycle. Animals were fed lettuce and fish food. For cell culture, snails 1 to 2 months old (10 mm-15 mm shell length) were used, whereas the brain Conditioned Medium (CM) was prepared from 2 to 3 months old snails (15 mm-25 mm shell length).
Cell Culture
Animals required for cell culture and CM preparation were deshelled with forceps and anesthetized for 7 minutes-10 minutes in 10% Listerine (21.0% ethanol, 0.042% menthol; Pfizer Canada, Toronto, Ontario, Canada) in normal Lymnaea saline (51.3 mM NaCl, 1.7 mM KCl, 4.0 mM CaCl2, 1.5 mM MgCl2), buffered to pH 7.9 with 2-hydroxyethyl piperazine-N’-2-ethanesulfonic acid (HEPES) [57-59]. All instruments required for dissections were sterilized with 70% ethanol. The animals were pinned down through their body wall to the bottom of the silicone rubber dissection dish (General Electric, RTV 616) containing antibiotic saline. An incision made through the dorsal midline with fine scissors, and the Central Ring Ganglia (CRG) was exposed by pinning down the body wall inside out. The esophagus, which passes through the CRG, was removed using fine scissors.
Extraction of Identified Neurons
Neurons were isolated individually and cultured, as described previously [58,59]. The central ring ganglia were isolated and then sterilized with three, fifteen-minute antibiotic washes (50 μg/ml gentamycin in normal saline). Ganglia were subsequently treated with trypsin (2 mg/ml, Sigma type III; Sigma Chemical Company, St. Louis, MO) for 23 minutes and then soybean trypsin inhibitor (2 mg/ml, Sigma type 1-S; Sigma Chemical Company) for 15 minutes, each dissolved in 50% L-15 (Gibco special order) Defined Medium (DM). DM was prepared with added inorganic salts (similar concentration as saline) and 20 μg/ml gentamycin, with the pH adjusted to 7.9 with 1N NaOH. Following enzymatic treatment, the ganglia were pinned to the bottom of a dissection dish containing high osmolarity DM (DM with added 20 mM D-glucose) and unsheathed with a pair of fine forceps. Suction pressure was applied through fire-polished, Sigmacoated pipettes (50-100 μm tip diameter) to remove desired neurons individually [60]. Isolated neurons were cultured on poly-L-lysine treated dishes either in the presence or absence of CM. The dishes were left undisturbed overnight.
Pressure Injection of Drugs
Chemicals. KN-93 (Calbiochem), KN-92 (Sigma) was first dissolved in dimethylsulfoxide (DMSO; Sigma). The stock solution (2 mM) was subsequently diluted in Lymnaea saline to make a final concentration of 100 nM immediately before the application. Final DMSO concentration in the diluted working solution was less than 0.01 % (DMSO<0.01%), and the drugs were pressure ejected (model 5242 (Eppendorf Scientific, Westbury, NY); 10-30-sec pulses, 6-10 psi, pipette tip diameter of 2-5 mm) directly onto the individual growth cones.
Images of growth cones were captured using a Contax (Toronto, Canada) camera mounted on a Zeiss (Oberkochen, Germany) Axiovert 135 inverted microscope. Growth cones were visually scored for a collapsed morphology.
Statistics
Data expressed as Mean ± Standard Errors of the Mean (SEM). Statistical analysis of the data conducted by using SPSS (IBM SPSS statistics 25) and graphs was constructed with Microsoft Office Excel 2019. The significant difference between various groups was determined by a t-test. Statistical significance was assumed if the p-value was less than 0.05 (*p<0.05).
Result
CaMKII is involved in Maintaining the Morphology of Neurite Growth Cone
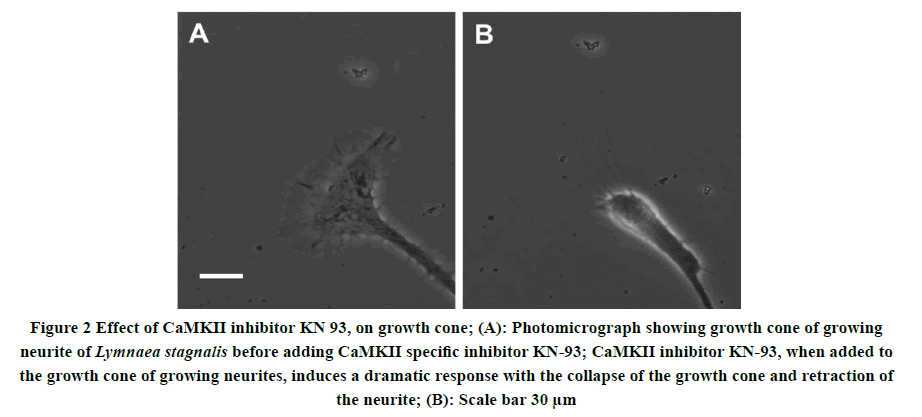
To determine whether Ca2+/calmodulin-dependent protein kinase activity is necessary to maintain the structure and function of the neurite growth cone of isolated molluscan neurons. Individually identified Pedal A (PeA) neurons were isolated from the Lymnaea CNS and plated on poly-L-lysine coated dishes containing CM and left undisturbed overnight. Neurite outgrowth was monitored and visualized under the microscope 24 hours-48 hours after initial plating. All neurons following neurite outgrowth have examined the effect of CaMKII inhibitor on growth cone structure and motility by using a CaMKII specific inhibitor KN-93. KN-93 (2 μl; 100 nM) was pressure applied directly onto individual growth cones. Figure 2 shows the structure of neuronal growth cones from intact Pedal A, neurons cultured in 2 ml of CM, before and after the application of CaMKII inhibitor KN-93. Within a few minutes of KN-93 application, the growth cone not only halts the motility but also exhibited collapse morphology and growth cone retraction in cultured neurons.
Figure 2. Effect of CaMKII inhibitor KN 93, on growth cone; (A): Photomicrograph showing growth cone of growing neurite of Lymnaea stagnalis before adding CaMKII specific inhibitor KN-93; CaMKII inhibitor KN-93, when added to the growth cone of growing neurites, induces a dramatic response with the collapse of the growth cone and retraction of the neurite; (B): Scale bar 30 μm
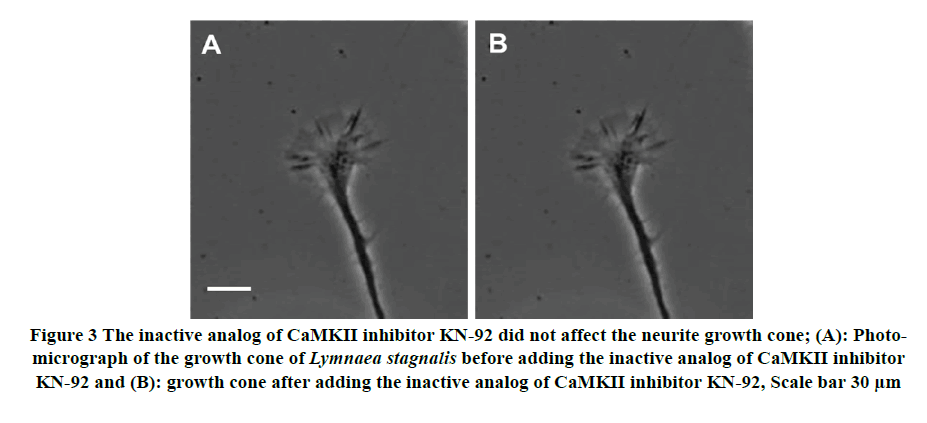
To test for the specificity of CaMKII inhibitor KN-93 effects on growth cone morphology, we used the inactive analog KN-92 (2 μl; 100nM) to see its effects on growth cone morphology. Despite multiple applications of KN-92 on the growth cone, the growth cone morphology and motility remain unchanged, suggesting that the KN-92 did not affect the structure of the cytoskeleton in the growth cone (Figure 3). These results indicate that: a) CaMKII specific inhibitor KN-93 indeed perturb the morphology of the growth cone in the Lymnaea model, and b) that CaMKII is necessary to maintain the structure of the cytoskeleton of the growth cone.
Figure 3. The inactive analog of CaMKII inhibitor KN-92 did not affect the neurite growth cone; (A): Photomicrograph of the growth cone of Lymnaea stagnalis before adding the inactive analog of CaMKII inhibitor KN-92 and (B): growth cone after adding the inactive analog of CaMKII inhibitor KN-92, Scale bar 30 μm
Effect of CaMKII Inhibitor on Neurite Growth Cone Motility Rate
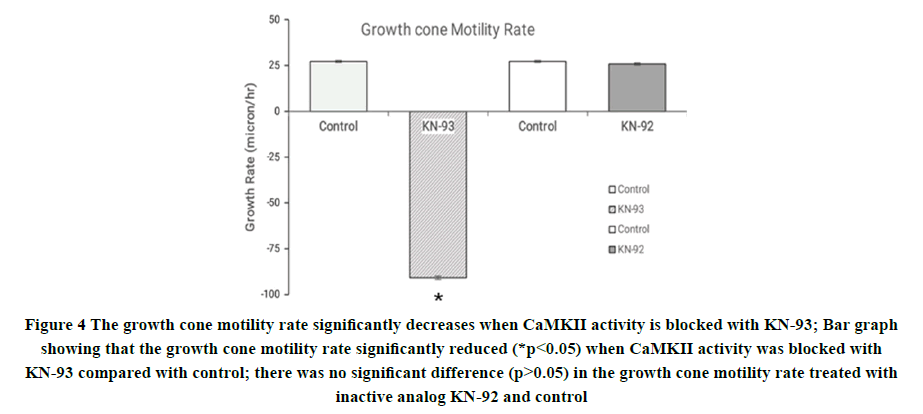
To determine the effect of CaMKII on the growth cone motility rate of the neurite growth cone, the identified Pedal A (PeA) neurons from the central ring ganglia of the freshwater pond snail Lymnaea stagnalis were cultured for 24 hrs-48 hrs in a 2 ml brain conditioned medium. To investigate the involvement of CaMKII in motility rate and growth cone advancement, we pressure applied CaMKII inhibitor (final concentration of DMSO<0.01%), KN-93 (2 μl; 100 nM in DMSO) directly onto individual growth cones. The growth cone motility rate of the growth cone was monitored before and after the applications of drugs. Within 20 minutes of its application, the growth cone motility rate was significantly reduced (p<0.05) (Figure 4). Specifically, the control growth cone motility rate was 27 ± 0.57 μm/h (n=10), which was reduced significantly in the presence of pressure applied KN-93 to -91 ± 0.93 μm/h (n=10; Figure 4). To test the specificity of KN-93 effects and control for the carrier solution (DMSO), we used the inactive analog KN-92 (also 100 nM). KN-92 or DMSO alone did not affect the growth cone motility rate, (control motility rate 27 ± 0.57 nm/h (n=10); KN-92 26± 0.61 nm/h (n=10; Figure 4)). These results indicate that CaMKII specific drugs indeed perturb its function in the Lymnaea model and that CaMKII is involved in controlling the growth cone motility rate.
Figure 4. The growth cone motility rate significantly decreases when CaMKII activity is blocked with KN-93; Bar graph showing that the growth cone motility rate significantly reduced (*p<0.05) when CaMKII activity was blocked with KN-93 compared with control; there was no significant difference (p>0.05) in the growth cone motility rate treated with inactive analog KN-92 and control
Discussion
In the present study, we have demonstrated that inhibition of CaMKII activity in the growth cone, with its specific inhibitor KN-93, dramatically changes the morphology and motility of the growth cone in identified cultured neurons. In addition, growth cones of neurons treated with KN-93 exhibit collapsed morphology. Blocking of CaMKII activity alters the growth cone morphology through the changes in cytoskeleton protein. Moreover, the assembly of cytoskeleton elements is responsible for the advancement and elongation of the neurite growth cone. This finding indicates that CaMKII serves as a pivotal molecule to maintain the structure and function of the neurite growth cone present at the tip of the growing neurite.
Calcium ion (Ca2+) and calmodulin (CaM) are important signaling molecules that have been shown to play a significant role in a wide variety of neuronal functions, including synaptic plasticity, neurite outgrowth of axon and dendrites [35,36]. Ca2+ signaling is mediated through several Ca2+-binding proteins, including CaM [35,61]. CaM is a regulatory protein that modulates the activity of several signaling molecules such as Protein Kinase C (PKC), phosphorylase kinase, myosin light chain kinase, calcineurin, and family of Ca2+/calmodulin-dependent protein kinase (CaMK) [32].
Growth cones present at the tips of growing neurites are responsible for axonal pathfinding, target cell selection, and specific synapse formation [62,63]. The morphology and motility of the growth cone depend on Ca2+ and the stimulation of Ca2+ dependent protein kinases (CaMK) in developing neurons. Cytosolic free Ca2+ and Ca2+ influx into axoplasm appear to regulate growth cone behavior by activating different signaling cascades, including cAMP and CaMKII [43,56,64-66]. CaMKII is a member of the CaMK family, and it is highly expressed in the cytosol of developing neurons, including the growth cone. CaMKII is critically involved in synaptic plasticity in the brain. Neuronal activity and the Ca2+ influx activate the CaMKII [34,35].
The movement of filopodia-like extensions is a necessary step for neuronal growth cones to find new connection sites that can then advance into nascent synapses and mature into functional synaptic connections [67-70]. Intracellular Ca2+ plays an essential role in promoting growth cone [34,71,72]. Ca2+ controls cytoskeletal dynamics of the growth cone through the modulation of polymerization and depolymerization of cytoskeletal proteins, including microtubules and F-actin [33,73,74]. Previous studies have shown that motile growth cones have higher free Ca2+ levels than growth that stops growing [75-77]. A moderate level of intracellular Ca2+ is necessary to neurite outgrowth, and extension of growth cone, very high or very low intracellular Ca2+ level impairs the neurite growth and halts the growth cone extension at the tip of the neurite [34,64]. The moderate Ca2+ level of the active growth cone of Snail Helisoma is an estimated range of 200 nm-1000 nm, whereas the non-motile growth cone has a 30 nM-70 nM intracellular Ca2+ level [34]. Previous studies indicate that the overexpression of CaMKII in neuron culture increases filopodia motility, dendritic arborization, and spine density. CaMKII protein expresses in the neurite growth cone and interacts with F-actin proteins to regulate the stability of cytoskeletal elements in the growth cone [56,78-80].
Conclusion
In summary, our findings suggest that CaMKII activity is critically needed to maintain the growth cone morphology and for the advancement and elongation of the growth cone to regulate the motility rate. Together with previous studies, our finding underscores the importance of CaMKII activity to maintain the structure and motility of growth cone in neuronal cultured.
Declarations
Conflicts of Interest
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Acknowledgment
This work is supported by the grant (GRP-105-41) from King Khalid University, Abha Kingdom of Saudi Arabia. The authors also would like to special thanks to Dr. Naweed Syed and Wali Zaidi from the University of Calgary, Canada, for all their help and support.
References
- Rosner, Jack, et al. "The potential for cellular therapy combined with growth factors in spinal cord injury." Stem Cells International, Vol. 2012, 2012.
- Sofroniew, Michael V. "Dissecting spinal cord regeneration." Nature, Vol. 557, No. 7705, 2018, pp. 343-50.
- Ibarretxe, Gaskon, et al. "Fast regulation of axonal growth cone motility by electrical activity." Journal of Neuroscience, Vol. 27, No. 29, 2007, pp. 7684-95.
- Kotov, Nicholas A., et al. "Nanomaterials for neural interfaces." Advanced Materials, Vol. 21, No. 40, 2009, pp. 3970-4004.
- Giger, Roman J., Edmund R. Hollis, and Mark H. Tuszynski. "Guidance molecules in axon regeneration." Cold Spring Harbor perspectives in Biology, Vol. 2, No. 7, 2010, p. a001867.
- Cunha, Carla, Silvia Panseri, and Stefania Antonini. "Emerging nanotechnology approaches in tissue engineering for peripheral nerve regeneration." Nanomedicine: Nanotechnology, Biology and Medicine, Vol. 7, No. 1, 2011, pp. 50-59.
- Assinck, Peggy, et al. "Cell transplantation therapy for spinal cord injury." Nature Neuroscience, Vol. 20, No. 5, 2017, pp. 637-47.
- Rosenzweig, Ephron S., et al. "Chondroitinase improves anatomical and functional outcomes after primate spinal cord injury." Nature Neuroscience, Vol. 22, No. 8, 2019, pp. 1269-75.
- Diaz-Galindo, Maria del Carmen, et al. "Therapeutic approaches of trophic factors in animal models and in patients with spinal cord injury." Growth Factors, Vol. 38, No. 1, 2020, pp. 1-15.
- Griffin, Jarred M., and Frank Bradke. "Therapeutic repair for spinal cord injury: Combinatory approaches to address a multifaceted problem." EMBO Molecular Medicine, Vol. 12, No. 3, 2020, p. e11505.
- Yang, Biao, et al. "Strategies and prospects of effective neural circuits reconstruction after spinal cord injury." Cell Death & Disease, Vol. 11, No. 6, 2020, pp. 1-14.
- Hassan, Atiq, Nazim Nasir, and Khursheed Muzammil. "Treatment strategies to promote regeneration in experimental spinal cord injury models." Neurochemical Journal, Vol. 15, No. 1, 2021, pp. 1-7.
- Heo, Chaejeong, et al. "The control of neural cell-to-cell interactions through non-contact electrical field stimulation using graphene electrodes." Biomaterials, Vol. 32, No. 1, 2011, pp. 19-27.
- Senger, J. L. B., et al. "Electrical stimulation as a conditioning strategy for promoting and accelerating peripheral nerve regeneration." Experimental Neurology, Vol. 302, 2018, pp. 75-84.
- Pockett, S., and R. M. Gavin. "Acceleration of peripheral nerve regeneration after crush injury in rat." Neuroscience Letters, Vol. 59, No. 2, 1985, pp. 221-24.
- Carmel, Jason B., and John H. Martin. "Motor cortex electrical stimulation augments sprouting of the corticospinal tract and promotes recovery of motor function." Frontiers in Integrative Neuroscience, Vol. 8, 2014, p. 51.
- Goganau, Ioana, et al. "Depolarization and electrical stimulation enhance in vitro and in vivo sensory axon growth after spinal cord injury." Experimental Neurology, Vol. 300, 2018, pp. 247-58.
- Choi, Elliot H., et al. "Epidural electrical stimulation for spinal cord injury." Neural Regeneration Research, Vol. 16, No. 12, 2021, pp. 2367-75.
- Zou, Yuanwen, et al. "Fabrication of aligned conducting PPy-PLLA fiber films and their electrically controlled guidance and orientation for neurites." ACS Applied Materials & Interfaces, Vol. 8, No. 20, 2016, pp. 12576-82.
- Fu, Chuan, et al. "Effect of electrical stimulation combined with graphene-oxide-based membranes on neural stem cell proliferation and differentiation." Artificial Cells, Nanomedicine, and Biotechnology, Vol. 47, No. 1, 2019, pp. 1867-76.
- Kotwal, Arundhati, and Christine E. Schmidt. "Electrical stimulation alters protein adsorption and nerve cell interactions with electrically conducting biomaterials." Biomaterials, Vol. 22, No. 10, 2001, pp. 1055-64.
- He, Zuhong, et al. "The structural development of primary cultured hippocampal neurons on a graphene substrate." Colloids and Surfaces B: Biointerfaces, Vol. 146, 2016, pp. 442-51.
- Kim, Tae-Hyung, Ki-Bum Lee, and Jeong-Woo Choi. "3D graphene oxide-encapsulated gold nanoparticles to detect neural stem cell differentiation." Biomaterials, Vol. 34, No. 34, 2013, pp. 8660-70.
- Hitscherich, Pamela, et al. "Electroactive graphene composite scaffolds for cardiac tissue engineering." Journal of Biomedical Materials Research Part A, Vol. 106, No. 11, 2018, pp. 2923-33.
- Sun, Yuan, et al. "Enhanced nerve cell proliferation and differentiation on electrically conductive scaffolds embedded with graphene and carbon nanotubes." Journal of Biomedical Materials Research Part A, Vol. 109, No. 2, 2021, pp. 193-206.
- Wang, Yu, et al. "Fluorinated graphene for promoting neuro‐induction of stem cells." Advanced Materials, Vol. 24, No. 31, 2012, pp. 4285-90.
- Guo, Weibo, et al. "Graphene microfiber as a scaffold for regulation of neural stem cells differentiation." Scientific Reports, Vol. 7, No. 1, 2017, pp. 1-8.
- Li, Ning, et al. "The promotion of neurite sprouting and outgrowth of mouse hippocampal cells in culture by graphene substrates." Biomaterials, Vol. 32, No. 35, 2011, pp. 9374-82.
- Tang, Mingliang, et al. "Enhancement of electrical signaling in neural networks on graphene films." Biomaterials, Vol. 34, No. 27, 2013, pp. 6402-11.
- Ghosh-Roy, Anindya, et al. "Calcium and cyclic AMP promote axonal regeneration in Caenorhabditis elegans and require DLK-1 kinase." Journal of Neuroscience, Vol. 30, No. 9, 2010, pp. 3175-83.
- Naoki, Honda, Yuichi Sakumura, and Shin Ishii. "Local signaling with molecular diffusion as a decoder of Ca2+ signals in synaptic plasticity." Molecular Systems Biology, Vol. 1, No. 1, 2005.
- Xia, Zhengui, and Daniel R. Storm. "The role of calmodulin as a signal integrator for synaptic plasticity." Nature Reviews Neuroscience, Vol. 6, No. 4, 2005, pp. 267-76.
- Goshima, Y., S. Ohsako, and T. Yamauchi. "Overexpression of Ca2+/calmodulin-dependent protein kinase II in Neuro2a and NG108-15 neuroblastoma cell lines promotes neurite outgrowth and growth cone motility." Journal of Neuroscience, Vol. 13, No. 2, 1993, pp. 559-67.
- Henley, John, and Mu-ming Poo. "Guiding neuronal growth cones using Ca2+ signals." Trends in Cell Biology, Vol. 14, No. 6, 2004, pp. 320-30.
- Zalcman, Gisela, Noel Federman, and Arturo Romano. "CaMKII isoforms in learning and memory: Localization and function." Frontiers in Molecular Neuroscience, Vol. 11, 2018, p. 445.
- Moro, Alessandro, et al. "CaMKII controls neuromodulation via neuropeptide gene expression and axonal targeting of neuropeptide vesicles." PLoS Biology, Vol. 18, No. 8, 2020, p. e3000826.
- Hudmon, Andy, and Howard Schulman. "Neuronal Ca2+/calmodulin-dependent protein kinase II: the role of structure and autoregulation in cellular function." Annual Review of Biochemistry, Vol. 71, No. 1, 2002, pp. 473-510.
- Meyer, Tobias, et al. "Calmodulin trapping by calcium-calmodulin-dependent protein kinase." Science, Vol. 256, No. 5060, 1992, pp. 1199-202.
- Braun, Andrew P., and Howard Schulman. "The multifunctional calcium/calmodulin-dependent protein kinase: From form to function." Annual Review of Physiology, Vol. 57, No. 1, 1995, pp. 417-45.
- Donai, Hitomi Fujishiro. "Study on the regulation of synaptic function by Ca2+/calmodulin-dependent protein kinase II." Yakugaku zasshi: Journal of the Pharmaceutical Society of Japan, Vol. 126, No. 5, 2006, pp. 337-42.
- Gordon‐Weeks, Phillip R. "Microtubules and growth cone function." Journal of Neurobiology, Vol. 58, No. 1, 2004, pp. 70-83.
- Lowery, Laura Anne, and David Van Vactor. "The trip of the tip: Understanding the growth cone machinery." Nature Reviews Molecular Cell Biology, Vol. 10, No. 5, 2009, pp. 332-43.
- Gasperini, Robert J., et al. "How does calcium interact with the cytoskeleton to regulate growth cone motility during axon pathfinding?" Molecular and Cellular Neuroscience, Vol. 84, 2017, pp. 29-35.
- Raper, Jonathan, and Carol Mason. "Cellular strategies of axonal pathfinding." Cold Spring Harbor perspectives in Biology, Vol. 2, No. 9, 2010, p. a001933.
- Goodhill, Geoffrey J., et al. "The dynamics of growth cone morphology." BMC Biology, Vol. 13, No. 1, 2015, pp. 1-18.
- Song, Hong-jun, and Mu-ming Poo. "The cell biology of neuronal navigation." Nature Cell Biology, Vol. 3, No. 3, 2001, pp. E81-88.
- Dickson, Barry J. "Molecular mechanisms of axon guidance." Science, Vol. 298, No. 5600, 2002, pp. 1959-64.
- Dent, Erik W., Stephanie L. Gupton, and Frank B. Gertler. "The growth cone cytoskeleton in axon outgrowth and guidance." Cold Spring Harbor perspectives in Biology, Vol. 3, No. 3, 2011, p. a001800.
- Ye, Xiyue, et al. "A subtle network mediating axon guidance: intrinsic dynamic structure of growth cone, attractive and repulsive molecular cues, and the intermediate role of signaling pathways." Neural Plasticity, Vol. 2019, 2019.
- Kolodkin, Alex L., and Marc Tessier-Lavigne. "Mechanisms and molecules of neuronal wiring: A primer." Cold Spring Harbor perspectives in Biology, Vol. 3, No. 6, 2011, p. a001727.
- Kerstein, Patrick C., Robert H. Nichol IV, and Timothy M. Gomez. "Mechanochemical regulation of growth cone motility." Frontiers in cellular neuroscience, Vol. 9, 2015, p. 244.
- Leong, Christopher CW, Naweed I. Syed, and Fritz L. Lorscheider. "Retrograde degeneration of neurite membrane structural integrity of nerve growth cones following in vitro exposure to mercury." Neuroreport, Vol. 12, No. 4, 2001, pp. 733-37.
- Sabry, James H., et al. "Microtubule behavior during guidance of pioneer neuron growth cones in situ." The Journal of Cell Biology, Vol. 115, No. 2, 1991, pp. 381-95.
- O'connor, Timothy P., and David Bentley. "Accumulation of actin in subsets of pioneer growth cone filopodia in response to neural and epithelial guidance cues in situ." The Journal of Cell Biology, Vol. 123, No. 4, 1993, pp. 935-48.
- Tanaka, Elly, and James Sabry. "Making the connection: Cytoskeletal rearrangements during growth cone guidance." Cell, Vol. 83, No. 2, 1995, pp. 171-76.
- Xi, Feng, et al. "Calcium/calmodulin‐dependent protein kinase II regulates mammalian axon growth by affecting F‐actin length in growth cone." Journal of Cellular Physiology, Vol. 234, No. 12, 2019, pp. 23053-65.
- Syed, N. I., A. G. Bulloch, and K. Lukowiak. "In vitro reconstruction of the respiratory central pattern generator of the mollusk Lymnaea." Science, Vol. 250, No. 4978, 1990, pp. 282-85.
- Woodin, Melanie A., David W. Munno, and Naweed I. Syed. "Trophic factor-induced excitatory synaptogenesis involves postsynaptic modulation of nicotinic acetylcholine receptors." Journal of Neuroscience, Vol. 22, No. 2, 2002, pp. 505-14.
- Munno, David W., David J. Prince, and Naweed I. Syed. "Synapse number and synaptic efficacy are regulated by presynaptic cAMP and protein kinase A." Journal of Neuroscience, Vol. 23, No. 10, 2003, pp. 4146-55
- Ridgway, R. L., et al. "Nerve Growth Factor (NGF) induces sprouting of specific neurons of the snail, Lymnaea stagnalis." Journal of Neurobiology, Vol. 22, No. 4, 1991, pp. 377-90.
- Khan, Shahid, Kenneth H. Downing, and Justin E. Molloy. "Architectural dynamics of CaMKII-actin networks." Biophysical Journal, Vol. 116, No. 1, 2019, pp. 104-19.
- Lockerbie, R. O. "The neuronal growth cone: A review of its locomotory, navigational and target recognition capabilities." Neuroscience, Vol. 20, No. 3, 1987, pp. 719-29.
- Kater, S. B., and Annemarie Shibata. "The unique and shared properties of neuronal growth cones that enable navigation and specific pathfinding." Journal of Physiology-Paris, Vol. 88, No. 3, 1994, pp. 155-63.
- Kater, S. B., and L. R. Mills. "Regulation of growth cone behavior by calcium." Journal of Neuroscience, Vol. 11, No. 4, 1991, pp. 891-99.
- Zheng, James Q., et al. "Turning of nerve growth cones induced by neurotransmitters." Nature, Vol. 368, No. 6467, 1994, pp. 140-44.
- Ming, Guo-li, et al. "cAMP-dependent growth cone guidance by netrin-1." Neuron, Vol. 19, No. 6, 1997, pp. 1225-35.
- Vaughn, James E. "Fine structure of synaptogenesis in the vertebrate central nervous system." Synapse, Vol. 3, No. 3, 1989, pp. 255-85.
- Jontes, James D., and Stephen J. Smith. "Filopodia, spines, and the generation of synaptic diversity." Neuron, Vol. 27, No. 1, 2000, pp. 11-14.
- Wong, Wai T., and Rachel OL Wong. "Rapid dendritic movements during synapse formation and rearrangement." Current Opinion in Neurobiology, Vol. 10, No. 1, 2000, pp. 118-24.
- Ahmari, Susanne E., and Stephen J. Smith. "Knowing a nascent synapse when you see it." Neuron, Vol. 34, No. 3, 2002, pp. 333-36.
- Gomez, Timothy M., and Nicholas C. Spitzer. "Regulation of growth cone behavior by calcium: New dynamics to earlier perspectives." Journal of Neurobiology, Vol. 44, No. 2, 2000, pp. 174-83.
- Gomez, Timothy M., et al. "Filopodial calcium transients promote substrate-dependent growth cone turning." Science, Vol. 291, No. 5510, 2001, pp. 1983-87.
- Yamauchi, Takashi, and Hitoshi Fujisawa. "Purification and cahracyterization of the brain calmodulin‐dependent protein kinase (kinase II), which is involved in the activtion of tryptophan 5‐monooxygnase." European Journal of Biochemistry, Vol. 132, No. 1, 1983, pp. 15-21.
- Gomez, Timothy M., and Paul C. Letourneau. "Actin dynamics in growth cone motility and navigation." Journal of Neurochemistry, Vol. 129, No. 2, 2014, pp. 221-34.
- Cohan, Christopher S., John A. Connor, and Stanley B. Kater. "Electrically and chemically mediated increases in intracellular calcium in neuronal growth cones." Journal of Neuroscience, Vol. 7, No. 11, 1987, pp. 3588-99.
- Mattson, Mark P., and Stanley B. Kater. "Calcium regulation of neurite elongation and growth cone motility." Journal of Neuroscience, Vol. 7, No. 12, 1987, pp. 4034-43.
- Goldberg, Daniel J. "Local role of Ca2+ in formation of veils in growth cones." Journal of Neuroscience, Vol. 8, No. 7, 1988, pp. 2596-605.
- Fink, Charles C., et al. "Selective regulation of neurite extension and synapse formation by the β but not the α isoform of CaMKII." Neuron, Vol. 39, No. 2, 2003, pp. 283-97.
- Easley, Charles A., et al. "Laminin activates CaMK-II to stabilize nascent embryonic axons." Brain Research, Vol. 1092, No. 1, 2006, pp. 59-68.
- Lin, Yu-Chih, and Lori Redmond. "Neuronal CaMKII acts as a structural kinase." Communicative & Integrative Biology, Vol. 2, No. 1, 2009, pp. 40-41.