Research - International Journal of Medical Research & Health Sciences ( 2023) Volume 12, Issue 6
Serum Interleukin-6 as a Potential Indicator of Severity in Covid-19 Patients: A Cohort Study
Naval Kishor Lodha1*, Krishna Murari2, Biramchand Mewara3, Gopal Sharma4 and Mahendra Verma52Associate Professor, Department of Biochemistry, Jhalawar Medical College, Jhalawar, Rajasthan, India
3Professor, Department of Surgery, Jhalawar Medical College, Jhalawar, Rajasthan, India
4Principal of Swasthye Kaliyan Paramedical College Jaipur, Rajasthan, India
5Principal of NIMS Paramedical College Jaipur, Rajasthan, India
Naval Kishor Lodha, Head of Department Medical Microbiology, Bhagwan Dhanwantri Chikitsa Seva Sansthan, Jhalawar, Rajasthan, India, Email: drnkjwr@gmail.com
Received: 12-Jun-2023, Manuscript No. ijmrhs-23-102091; Editor assigned: 14-Jun-2023, Pre QC No. ijmrhs-23-102091(PQ); Reviewed: 22-Jun-2023, QC No. ijmrhs-23-102091(Q); Revised: 25-Jun-2023, Manuscript No. ijmrhs-23-102091(R); Published: 30-Jun-2023
Abstract
Interleukin-6 is produced by a variety of cell types, but the most important sources are macrophages and monocytes at inflammatory sites. IL-6 is the chief stimulator for the production of most acute-phase proteins, whereas the other implicated cytokines influence subgroups of acute-phase proteins. IL-6 stimulates the inflammatory and auto-immune processes in many diseases such as multiple sclerosis, Neuro Myelitis Optica Spectrum Disorder (NMOSD), diabetes, atherosclerosis, depression, Alzheimer's disease, systemic lupus erythematosus, multiple myeloma, prostate cancer, Behçet's disease, rheumatoid arthritis, and intracerebral hemorrhage.
Hence, there is an interest in developing anti-IL-6 agents as therapy against many of these diseases. The first such is tocilizumab, which has been approved for rheumatoid arthritis. Castleman's disease and systemic juvenile idiopathic arthritis. Others are in clinical trials
Keywords
Interleukin-6, Neuro Myelitis Optica Spectrum Disorder (NMOSD), Covid-19, Case study
Introduction
The outbreak of coronavirus disease 2019 (COVID-19) in December 2019, was caused by the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). It poses a severe threat to global public health. Recently in the ongoing pandemic, Interleukin-6 (IL-6) test has been a frequently ordered test to evaluate and monitor the immune reaction in COVID-19 patients. IL-6 is a pleiotropic cytokine with a wide range of functions. IL-6 is produced from a single gene encoding a product of 212 amino acids, which is cleaved at the N terminus to produce a 184 amino acids peptide with a molecular weight between 22 kDa-27 kDa. IL-6 production is rapidly induced in the course of the acute inflammatory response associated with injury, trauma, stress, infection, brain death, neoplasia, and other conditions. IL-6 is produced by fibroblasts, activated T cells, activated monocytes or macrophages, and endothelial cells. IL-6 is a pro-inflammatory cytokine, that is secreted into the serum and induces a transcriptional inflammatory response through interleukin-6 receptor, alpha. IL-6 is a non-specific marker and hence is not diagnostic for any specific disease. IL-6 was proposed to be associated with the severity of coronavirus disease [1- 4].
Objectives
To correlate the serum level of IL-6 with the severity of COVID-19 and monitoring of the disease in adult COVID19 patients.
Methodology
A Cohort study was carried out after obtaining approval from the relevant Ethics Committee. A nasopharyngeal swab was taken as a sample for RT-PCR. This study was conducted first time in the Hadoti region of Jhalawar, Rajasthan. In this study, we have divided patients into three groups common (N=30), severe (N=30), and critical (N=30) cases of Covid-19 patients. The diagnosis and severity classification was based on clinical guidance for the management of adult Covid-19 patients (Ministry of Health & Family Welfare) and WHO interim guideline [5-8].
Common Cases
Covid-19 patients showing:
• Fever
• Dry cough
• Nasal congestion
• Sore throat
• Lost sense of smell or taste
• Conjunctivitis
• Mild fatigue
• Imaging showed pneumonia
Severe Cases
Adult cases meeting any of the following criteria:
• Respiratory distress (≥ 30 breaths/Min.).
• Oxygen saturation ≤ 93% at rest.
• The arterial partial pressure of Oxygen (PaO2)/ Fraction of inspired oxygen (FiO2) ≤ 300 mmHg.
• Chest imaging that shows obvious lesion progression within 24 hours.
Critical Cases
Cases meeting any of the following criteria:
• Respiratory failure and requiring mechanical ventilation.
• Shock.
• With other organ failures that require ICU care.
Diagnosis of Cases
Etiological: A positive RT-PCR detection of 2019-n CoV Nucleic acid.
Serological: The 2019-nCoV- Specific IgM&IgG antibody tests +ve.
Quantitative estimation of IL-6 is done by fully automated, chemiluminescent immunoassay in serum.
Biostatistics
• Descriptive statistics were applied to summarize data.
• Results were reported as mean with standard deviation.
• One-way ANOVA was applied to detect significant differences among stratifications.
• Statistical Package for Social Science (SPSS) 25.0 software was used for statistical analysis and illustration.
Results
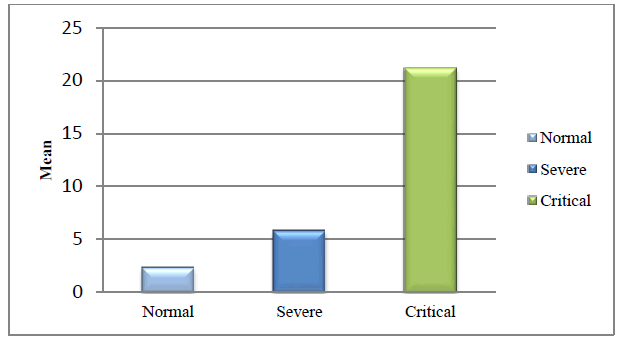
The mean concentration of IL-6 was 2.36 pg/ml ± 0.64 pg/ml, 5.84 pg/ml ± 1.12 pg/ml, and 21.21 pg/ml ± 6.57 pg/ml for the common, severe, and critical groups respectively (p<0.001). Our result shows that serum IL-6 levels increased as the disease progressed toward critical condition (Table 1 and Figure 1)
| Covid-19 Group | Number | Mean IL-6 (± S.D.) | p-value | F-Value |
|---|---|---|---|---|
| Normal | 30 | 2.36 ± 0.64 | <0.0001 | 201.85 |
| Severe | 30 | 5.84 ± 1.12 | <0.0001 | |
| Critical | 30 | 21.21 ± 6.57 | <0.0001 |
DISCUSSION
Serum IL-6 concentration was tested and analyzed in all groups of Covid-19 patients. We obtain a consistently high level of IL-6 in a severe and critical group of patients as compared to a normal group of Covid-19 patients. It is recognized that excessive, multifunctional host immune response may play an important role in the development and maintenance of the critical stage of Covid-19.
Conclusions
Serum IL-6 should be included in diagnostic workup to stratify disease severity. Therefore, full monitoring of the severity of COVID-19 and effective early intervention are the fundamental measures for reducing mortality
Declarations
Conflicts of Interest
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Future Scope of this Study
Our research work will help make novel strategies for the diagnosis, treatment, and prognosis of Covid-19 patients. This study may be helpful to reduce mortality due to Covid-19.
References
- Wikipedia: Interleukin 6. "GRCh38: Ensembl release 89: ENSG00000136244." Ensembl, 2017
- Wikipedia: Interleukin 6. "GRCm38: Ensembl release 89: ENSMUSG00000025746." Ensembl, 2017.
- National Center for Biotechnology Information, U.S. "Human PubMed Reference." National Library of Medicine.
- National Center for Biotechnology Information, U.S."Mouse PubMed Reference." National Library of Medicine.
- Ferguson-Smith, Anne C., et al. "Regional localization of the interferon-β2B-cell stimulatory factor 2/hepatocyte stimulating factor gene to human chromosome 7p15-p21." Genomics, Vol. 2, No. 3, 1988, 203-08.
Google Scholar Crossref - "Raised troponin and interleukin-6 levels are associated with a poor prognosis in COVID-19. " Cardiac Rhythm News, 2020.
- Banks, William A., Abba J. Kastin, and Enrique G. Gutierrez. "Penetration of interleukin-6 across the murine blood-brain barrier." Neuroscience letters, Vol. 179, No. 1-2, 1994, pp. 53-56.
Google Scholar Crossref - Wernstedt, Ingrid, et al. "Reduced stress-and cold-induced increase in energy expenditure in interleukin-6-deficient mice." American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, Vol. 291, No. 3, 2006, pp. 551-57.
Google Scholar Crossref