Research - International Journal of Medical Research & Health Sciences ( 2021) Volume 10, Issue 4
Study of Hospital Acquired Methicillin-Resistant Staphylococcus aureus in a Tertiary Care Hospital of Bihar
Suprakash Das1*, Keshav Bimal2, Namrata Kumari2 and Shailesh Kumar22Department of Microbiology, IGIMS, Patna, India
Suprakash Das, Department of Microbiology, HIMS, Barabanki, India, Email: suprakashdas@gmail.com
Abstract
Objective: There has been a steady increase in the prevalence of Methicillin-Resistant Staphylococcus Aureus (MRSA) infections throughout the world, especially in hospital settings, and is exacerbated by its ability to cause cross infections, colonization, and resistance to multiple classes of commonly used antimicrobials. There are various risk factors associated with MRSA infections among hospitalized patients. Studies which focus on current antimicrobial susceptibility pattern and risk factors associated with such important pathogen are scanty for this region and will be of great importance in the prevention and proper antimicrobial therapy for such infections.
Methods: Patients admitted to various departments of Indira Gandhi Institute of Medical Sciences, Patna in the duration of February 2014 to November 2015 were included in the study. Staphylococcus aureus and further MRSA were identified using the standard method according to CLSI 2014 guidelines. Isolates screened as MRSA were further tested for identification and antimicrobial susceptibility testing using VITEK-2 Compact (biomerieux, USA). Hospital-acquired MRSA and Methicillin Sensitive Staphylococcus Aureus (MSSA) infections were analyzed for associated risk factors by collecting their data for various laboratory and clinical parameters. Chi-square test was used for comparison between various categorical variables between patients with hospital-acquired MSSA and MRSA infections where p ≤ 0.05 were considered statistically significant.
Results: Out of the 102 Staphylococcus aureus isolates, 27 (26.5%) were identified as MRSA strains and 75 (73.5%) were MSSA strains. Most of the MRSA isolates (66.7%) and MSSA isolates (62.7%) were isolated from patients below 40 years of age. There is a male predominance in both MRSA (M:F=1.1:1) and MSSA (M:F=1.3:1) isolates. Orthopedics and Surgery departments contributed to the majority of MRSA isolates (48.15%) while Medicine and Orthopedics contribute to most of the MSSA isolates (37.3%). Both the MRSA and MSSA strains were mostly isolated from pus (38.2%) and blood samples (23.5%). Vancomycin and Linezolid showed the best sensitivity against MRSA strains. Both the MRSA and MSSA strains were highly resistant to ampicillin and penicillin. Antibiotics like erythromycin, clindamycin, and gentamicin showed statistically significant (p ≤ 0.05) differences in resistance among MRSA and MSSA strains. Several risk factors like length of stay in hospital or ICUs, burn injury, severe trauma, catheterization, use of mechanical ventilators, etc. were found to be statistically significant (p ≤ 0.05) for MRSA infections.
Conclusion: MRSA, with its high prevalence, continues to be an important pathogen regarding healthcare-associated infections. This organism is also multidrug-resistant and selecting proper antimicrobials for empirical therapy should be based on the current antibiogram obtained in the region.
Keywords
MRSA, MSSA, HAIs, Prevalence, Risk factors, Antimicrobial resistance
Introduction
Staphylococcus aureus (S. aureus), a very common pathogen in clinical practice, causes a broad spectrum of diseases ranging from minor skin infections, osteomyelitis, food poisoning to pneumonia, toxic shock syndrome, wound infections, and bacteremia [1]. Methicillin-Resistant Staphylococcus aureus (MRSA) are strains of Staphylococcus aureus that are resistant to methicillin and a large group of Beta-lactam antibiotics which include penicillin and the cephalosporins [2]. MRSA emerged as a nosocomial pathogen in the early 1960s and resulted in many sporadic and large outbreaks in different hospitals throughout the world. By the 1970s MRSA was endemic in many hospitals but in the 1990s it unexpectedly spread rapidly in communities to become Community Associated MRSA (CA-MRSA) [3,4]. An MRSA isolate is classified as ‘Hospital-Acquired MRSA’ (HA-MRSA) if it is isolated from a patient who has given a positive culture taken more than 48 hours after hospital admission and treated in the inpatient setting [5]. Bacteremia, surgical wound infections, and pneumonia are the most common nosocomial infections caused by MRSA [6]. There has been a consistent increase in the prevalence of MRSA infections throughout the world, especially in hospital settings as shown in data by various studies and antimicrobial surveillance programs including the National Nosocomial Infections Surveillance System (NNISS) which reported an escalation of MRSA infections among large hospitals in the United States from 4% in the 1980s to 50% in the late 1990s [7,8]. The problem of the MRSA infection is augmented by its ability to cause cross infections, colonization, and resistance to multiple classes of commonly used antimicrobials [1,9]. Transmission of MRSA stains can occur by various routes including the hands of healthcare workers, contaminated surfaces, and instruments or even it may be airborne [10,11].
Compared with MSSA (Methicillin Susceptible Staphylococcus aureus) strains, infections caused by MRSA strains are associated with higher morbidity, mortality, and health care burden [12].
Studies conducted by various authors have found that various risk factors are significantly associated with acquisition and infection by MRSA and those risk factors can vary in different regions and health care setups [13,14].
Taking into consideration the fact that MRSA has become one of the most important pathogens in hospital and community settings. This study was undertaken to determine the prevalence and associated risk factors for MRSA infections which will be crucial to formulate infection control and prevention policies and to determine the antimicrobial susceptibility pattern of MRSA isolates which will guide the clinicians to construct proper antimicrobial therapy for such infections and reduce antibiotic misuse and its associated adverse effects including antimicrobial resistance.
Materials and Methods
After approval of the Institute Ethical Committee (Memo No: IGIMS/2014/260 Acad.), this cross-sectional study was conducted in the Department of Microbiology, I.G.I.M.S, Patna, Bihar, from February 2014 to November 2015.
Various clinical samples like pus; swabs (from wounds, eye, ear, throat, and urethra); body fluids (Ascitic fluid, Pleural fluid, Peritoneal fluid, Bile, Liver aspirates, CSF); tissue samples, sputum, CV lines, catheter tips, shunts, ET Tubes, and urine, from patients only after 48 hours of admission in various departments, were included in this study.
Staphylococcus aureus was identified using the standard microbiological methods and MRSA by cefoxitin disc (30 μg) and agar screening media. Antimicrobial susceptibility tests were done on Mueller-Hinton agar with commercially available antibiotic discs (Hi-Media, Mumbai) according to CLSI 2014 guidelines (Figure 1-3) [15]. Isolates screened as MRSA were further tested for identification and antimicrobial susceptibility testing using VITEK-2 Compact (Biomérieux, USA).
Based on previous studies by various authors prevalence of HA-MRSA was assumed as 25% and with a precision of 10%, the sample size was estimated as a minimum of 73 [16]. Repeat samples from the same patient were not included in the study.
Analysis of Risk Factors
Demographic, laboratory, and clinical data of all the patients who were admitted for more than 48 hours and culture yields Staphylococcus aureus, was divided into two groups-patients with MRSA isolates (HA-MRSA) and patients with MSSA isolates (HA-MSSA). All the variables were recorded for both these 2 groups for statistical analysis.
Statistical Analysis
All the data were plotted in Microsoft Excel Sheet (v.2007). Descriptive statistics were used to describe characteristics of HA-MRSA and HA-MSSA cases. Associations between categorical variables were tested using Chi-square tests.
All statistical analyses were performed using MedCalc (v.19.1.3) and a p-value of ≤ 0.05 was considered statistically significant.
Results
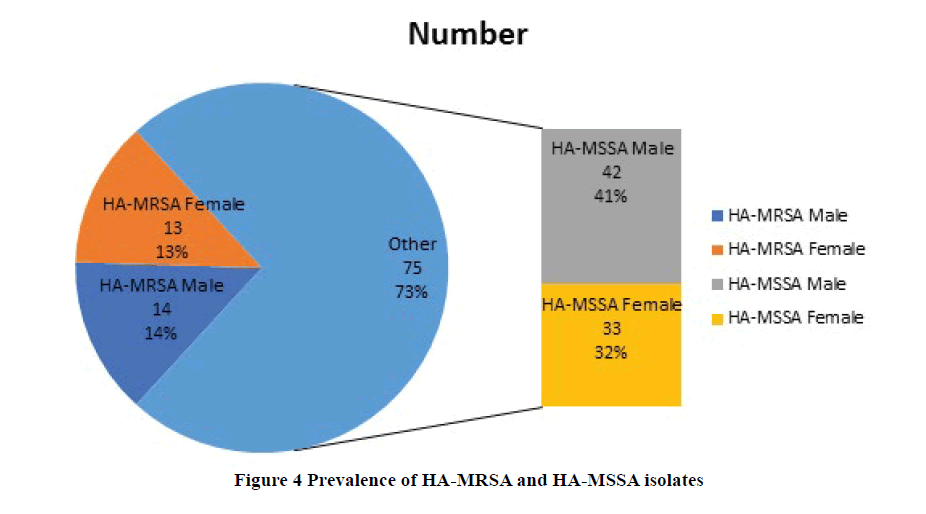
A total of 14182 clinical samples were received in the Microbiology laboratory from February 2014 to November 2015, out of which 102 hospital-acquired Staphylococcus aureus isolates were taken for further study. Out of the 102 Staphylococcus isolates, 27 (26.5%) were identified as MRSA strains and 75 (73.5%) were MSSA strains (Figure 4).
Of the 102 patients who had the Staphylococcal infection, 56 (54.9%) patients were male, and 46 (45.1%) were female with a male: female ratio of 1.2. Likewise, the majority of the patients with MRSA isolates were also male, 14/27 (51.85%), and male to female ratio for MRSA isolates, was 14:13 or approximately 1.1:1. In the case of patients with MSSA isolates, it was also the male patients who form the majority i.e. 42/75 (56%), and the male-to-female ratio was 42:33 or approximately 1.3:1 (Figure 1).
The majority of the patients from whom Staphylococcus aureus was isolated were in the age group of 0-40 years, 65/102 (63.7%). In the case of patients with isolation of HA-MRSA strains majority of the patients were of 0-40 years of age, 18/27 (66.7%) which was also seen in patients with isolation of HA-MSSA strains, 47/75 (62.7%) (Table 1, and Table 2).
| Age Group (Yrs) | Male (n=14) | Female (n=13) | Total (%) |
|---|---|---|---|
| 0-10 (n=4) | 2 | 2 | 10 (37%) (Pediatric age) |
| 11-20 (n=6) | 4 | 2 | |
| 21-30 (n=3) | 2 | 1 | 12 (44.4%) (Adults) |
| 31-40 (n=5) | 2 | 3 | |
| 41-50 (n=1) | 1 | 0 | |
| 51-60 (n=3) | 2 | 1 | |
| 61-70 (n=3) | 1 | 2 | 5 (18.5%) (Old) |
| 71-80 (n=2) | 0 | 2 | |
| Total | 14 | 13 | 27 |
| Age Group (Yrs) | Male (n=42) | Female (n=33) | Total (%) |
|---|---|---|---|
| 0-10 (n=14) | 8 | 6 | 22 (29.3%) (Pediatric age) |
| 11-20 (n=08) | 3 | 5 | |
| 21-30 (n=13) | 8 | 5 | 44 (58.7%) (Adults) |
| 31-40 (n=12) | 7 | 5 | |
| 41-50 (n=09) | 5 | 4 | |
| 51-60 (n=10) | 6 | 4 | |
| 61-70 (n=05) | 3 | 2 | 09 (12%) (Old) |
| 71-80 (n=04) | 2 | 2 | |
| Total | 42 | 33 | 75 |
| Departments (Including ICUs) | HA-MRSA (n=27) | HA-MSSA (n=75) | Total (%) |
|---|---|---|---|
| Pediatrics | 5 | 11 | 16 (15.7%) |
| Medicine | 5 | 15 | 20 (19.6%) |
| Surgery | 6 | 12 | 18 (17.6%) |
| Orthopedics | 7 | 13 | 20 (19.6%) |
| OBG | 2 | 8 | 10 (9.8%) |
| Urology | 1 | 6 | 7 (6.9%) |
| ENT | 0 | 3 | 3 (2.9%) |
| Ophthalmology | 0 | 2 | 2 (1.96%) |
| Dermatology | 1 | 5 | 6 (5.9%) |
| Total | 27 | 75 | 102 |
The majority of the 102 strains of Staphylococcus aureus were isolated from pus 38.2% (39/102) followed by blood 23.5% (24/102) and urine 16.7% (17/102). Likewise, 11 isolates (40.74%) of HA-MRSA strains were isolated from pus followed by 7 isolates (25.93%) from blood and 4 isolates (14.8%) from urine samples. In the case of HA-MSSA isolates, the majority of them were obtained from pus 37.3% (28/75) followed by blood 22.7% (17/75) and urine 17.3% (13/75) (Table 4).
| SPECIMENS | HA-MRSA (n=27) | HA-MSSA (n=75) | Total (%) |
|---|---|---|---|
| Pus | 11 | 28 | 39 (38.2%) |
| Urine | 4 | 13 | 17 (16.7%) |
| Blood | 7 | 17 | 24 (23.5%) |
| AF (Ascitic Fluid) | 1 | 2 | 3 (2.9%) |
| Central line tip | 2 | 3 | 5 (4.9%) |
| Sputum | 0 | 3 | 3 (2.9%) |
| HVS (High Vaginal Swabs) | 0 | 2 | 2 (1.96%) |
| CSF | 0 | 2 | 2 (1.96%) |
| PF (Pleural Fluid) | 0 | 2 | 2 (1.96%) |
| Swab | 2 | 3 | 5 (4.9%) |
| Total | 27 | 75 | 102 |
The majority of the HA-MSSA isolates showed resistance to Penicillin (98.7%) followed by Ampicillin (94.7%) and Nalidixic acid (72%). All the isolates were sensitive to Vancomycin and Linezolid followed by Co-trimoxazole (97.3%), Chloramphenicol (89.3%), and Amikacin (84%).
All the strains of HA-MRSA were resistant to Penicillin (100%), Ampicillin (100%) followed by Nalidixic acid (96.3%), Erythromycin (92.6%), and Gentamicin (85.2%). All the isolates were sensitive to Vancomycin and Linezolid (100%) followed by Co-trimoxazole (77.8%) and Amikacin (70.4%).
For urinary isolates, Nitrofurantoin showed 50% and 23.1% resistance for HA-MRSA and HA-MSSA isolates (Table 5).
| Antibiotics | HA-MRSA (resistant %) | HA-MSSA (resistant %) | p-value |
|---|---|---|---|
| Penicillin | 27/27 (100%) | 74/75 (98.7%) | NA |
| Ampicillin | 27/27 (100%) | 71/75 (94.7%) | NA |
| Erythromycin | 25/27 (92.6%) | 32/75 (42.7%) | <0.00001 (S) |
| Clindamycin | 21/27 (77.8%) | 19/75 (25.3%) | <0.00001 (S) |
| Amikacin | 8/27 (29.6%) | 12/75 (16%) | 0.126119 (NS) |
| Gentamicin | 23/27 (85.2%) | 16/75 (21.3%) | <0.00001 (S) |
| Nalidixic Acid | 3/27 (96.3%) | 54/75 (72%) | 0.8487 (NS) |
| Ciprofloxacin | 22/27 (81.5%) | 39/75 (52%) | 0.738 (NS) |
| Nitrofurantoin | 2/4 (50%) | 3/13 (23.1%) | .301409 (NS) |
| Tetracycline | 20/27 (74.1%) | 23/75 (30.7%) | 0.09 (NS) |
| Co-trimoxazole | 6/27 (22.2%) | 2/75 (2.7%) | 0.1191 (NS) |
| Chloramphenicol | 11/27 (40.7%) | 8/75 (10.7%) | 0.578 (NS) |
| Linezolid | 0/27 (0%) | 0/75 (0%) | NA |
| Vancomycin | 0/27 (0%) | 0/75 (0%) | NA |
S: Significant; NS: Not Significant; NA: Not Applied
Several risk factors were analyzed for association with HA-MRSA infections and nine of them were found to be statistically significant (p ≤ 0.05) and those risk factors are the length of stay in hospital or ICUs, burn injury, severe trauma, and presence of open wounds, catheterization, use of mechanical ventilators, use of antibiotics before admission and more than two previous hospital admissions (Table 6).
| Risk Factors | HA-MRSA (n=27) | HA-MSSA (n=75) | p-value |
|---|---|---|---|
| Age Groups | |||
| Pediatric age (≤ 18 years) | 10 | 22 | 0.46 (NS) |
| Adults | 12 | 44 | 0.20 (NS) |
| Older (≥ 60 yrs) | 5 | 9 | 0.40 (NS) |
| Total | 27 | 75 | |
| Sex | |||
| Male | 14 | 42 | 0.71 (NS) |
| Female | 13 | 33 | 0.71 (NS) |
| Total | 27 | 75 | |
| ≥1 Week of LOS before a positive culture | 17 | 23 | 0.003 (S) |
| Burn injury | 9 | 7 | 0.003 (S) |
| Diabetes Mellitus | 6 | 17 | 0.96 (NS) |
| Severe Trauma | 12 | 14 | 0.008 (S) |
| Open wounds and ulcers | 15 | 19 | .004 (S) |
| LOS in ICUs ≥ 1 week | 16 | 21 | 0.004 (S) |
| Malignancy | 4 | 7 | 0.43 (NS) |
| CKD | 6 | 11 | 0.37 (NS) |
| Antibiotics use (within 24 hours of admission) | 21 | 36 | 0.008 (S) |
| Catheterization | 18 | 27 | 0.006 (S) |
| Hemodialysis | 5 | 9 | 0.40 (NS) |
| Abnormal Liver Functions | 4 | 13 | 0.76 (NS) |
| ≥ 2 Previous hospitalizations up to admission | 17 | 21 | 0.001 (S) |
| Use of Mechanical ventilators | 11 | 9 | 0.001 (S) |
LOS: Length of Stay; CKD: Chronic Kidney Disease
Discussion
The prevalence of HA-MRSA in this study was 26.5% (27/102) which shows the significant role of MRSA as a nosocomial pathogen. Studies done by various authors have proved the importance of MRSA in a hospital environment. A pilot program of MRSA surveillance conducted in various centers across India in 1996 found 26.65% of Staphylococcal strains to be MRSA in Mumbai, 47.1% in Bangalore, and 42.5% in New Delhi [16]. Verma S, et al. in 2000, reported that the prevalence of MRSA in their center was as low as 12% in 1992 increasing to 80.89% in 1999 [17]. Other authors have reported diverse findings as far as isolation of MRSA is concerned viz. 54.85% from Uttar Pradesh (Anupurba, et al.), 31.1% from Tamil Nadu (Rajaduraipandi K, et al.), 44% from New Delhi (Tyagi A, et al.), and 38.14% from Sikkim (Tsering DC, et al.) [18-20].
Our finding corresponds to findings by other authors from different states of India where all have seen that MRSA has a significant prevalence in healthcare-associated infections.
In this study, patients from age groups consisting of pediatric and young adults contribute to the majority of cases (65, >63%) which is seen in the case of both HA-MRSA (66.7%) and HA-MSSA (62.7%) isolates. Studies conducted by Chen CJ, et al., and Eshwara VK, et al. have shown a similar age group consisting of children and young adults are the major contributors (>57%) of Staphylococcal cases including MRSA isolates [21,22]. A study conducted by Waness A, et al. however reported higher infection rates of MRSA in persons above 65 years of age [23]. This finding is quite different from the present study in which only 14 (13.7%) patients are there in the age group of 61 and above with isolation of HA-MRSA and HA-MSSA isolates.
In this study, there was a male predominance in cases of both HA-MRSA (M:F=1.1:1) and HA-MSSA (M:F=1.3:1) isolates. This may be due to the fact like differences in MRSA carriage in males as compared to females due to differences in hand hygiene and daily activities. A study conducted by Winkler J, et al. showed that female hormones like estrogen may have some role in limiting colonization, and in rural settings; females less readily seek medical care for their ailments [24]. A study conducted by Humphreys H, et al. showed that there is a male predominance (OR>1.3) in staphylococcal infections including MRSA infections which is in harmony with our study [25].
Most of the Staphylococcal isolates (74/102, 72.5%) which include both HA-MRSA and HA-MSSA were isolated from Pediatrics, Medicine, Surgery, and Orthopedics departments and ICUs corresponding to those departments. A study conducted by Severin, et al. reported a high prevalence of MRSA carriage in patients admitted in surgical wards [26]. A study conducted by Arora S, et al. also showed a similar finding where Orthopedics (75/250), Pediatrics (56/250), Medicine (44/250), Surgery (26/250), and Gynecology department (27/250) contributes to the majority of MRSA isolates [27]. The above studies including the current study established the fact that strict MRSA infection control practices are needed especially in those departments and surveillance for MRSA carriage may be a crucial step towards infection control and prevention.
Most of the Staphylococcal isolates (HA-MRSA and HA-MSSA) were isolated from pus, blood, and urine samples (80/102, 79.5%). Anupurva S, et al. reported maximum isolation (200/381, i.e. 52.5%) from pus and wound swabs [18]. Rajaduraipandi, et al. also reported that the majority of the MRSA isolates (>62%) were from blood and pus samples [19]. The findings of their studies correspond to our findings and emphasize the fact if the primary Gram stain from such samples showed the presence of Gram-positive cocci in clusters, then further tests for identification of MRSA should be carried out.
The antimicrobial susceptibility pattern of MRSA isolates showed that they are resistant to multiple classes of antibiotics and many of those isolates are resistant to more than three different classes of antibiotics which makes them important Multi-Drug Resistant Organisms (MDRO) regarding hospital-associated infections [28]. In this study, a very high resistance (>95%) is seen against beta-lactam antibiotics like Penicillin and Ampicillin and shouldn’t be used empirically whenever hospital-acquired staphylococcal infections are suspected. In this study, we have also found a statistically significant difference (p ≤ 0.05) among sensitivity shown by various antibiotics like Erythromycin, Clindamycin, Gentamicin, and Tetracyclin among HA-MRSA and HA-MSSA isolates. This infers that those antibiotics may show a good sensitivity when used against HA-MSSA isolates but may be ineffective when used against HAMRSA isolates and thus they should be only used after antimicrobial susceptibility testing. Vancomycin and Linezolid provide the best option against HA-MRSA and HA-MSSA isolates (100% sensitivity).
Anupurba S, et al. reported in a study that 80% of their MRSA strains were resistant to antibiotics like cephalexin, ciprofloxacin, penicillin, co-trimoxazole, gentamycin, erythromycin, and tetracycline [18]. A study conducted by Arora S, et al. showed that 73% of the MRSA strains were multidrug-resistant [27]. Although Vancomycin is still the empirical treatment of choice in severe MRSA infections in less severe conditions oral antibiotics should be preferably used depending on the antimicrobial susceptibility testing [29,30].
In this study, authors have analyzed various risk factors that may be associated with HA-MRSA infections in comparison to HA-MSSA infections based on previous studies conducted by various authors [13,14]. The authors have found that some of the risk factors were statistically significant (p ≤ 0.05) among patients with MRSA isolates. Length of stay (>1 week) in hospital wards and ICUs, burn injury, severe trauma and open wounds, use of broad-spectrum antibiotics at the time or before admission to hospital, catheterization, patients on mechanical ventilators, and patients having more than 2 previous admissions in hospitals are important risk factors for MRSA infections than MSSA infections.
A study conducted by Harinstein L, et al. showed that many risk factors like catheterization and more than 2 previous hospitalizations are associated with the conversion of MRSA colonization into MRSA infections [31].
A study conducted by Yao Z, et al. showed Length of Stay (LOS) in the hospital and the use of mechanical ventilation as important risk factors [32].
Another study by Yamakawa K. et al. showed that four risk factors which include intubation, presence of open wounds, antimicrobial therapy, and use of steroids if occurred within 24 hours of ICU admission are associated with MRSA infections [33].
Current study results are in harmony with findings of those above-mentioned authors whereas, in some risk factors like diabetes mellitus and liver cirrhosis, we don’t find statistical significance as compared to work done by Wu HP, et al. [34].
The knowledge of various risk factors may help in identifying susceptible patients for MRSA colonization or infections and active surveillance of those patients along with health care workers attending those patients may play a crucial role in controlling HA-MRSA infections and prevent associated mortality, morbidity, and economic burden of health care [35,36].
Conclusion
According to this study, MRSA, with its high prevalence, continues to be an important pathogen in Bihar. These organisms are multidrug-resistant and selecting proper antimicrobials for empirical therapy should be based on the current antimicrobial susceptibility pattern, the severity of the infection, and other associated risk factors of the patient. Active surveillance of patients based on their risk profile for colonization of MRSA may play an important role in the control and prevention of MRSA spread and related infections which will, in turn, reduce the associated mortality, morbidity, and cost of health care.
Limitations of the Study
In this study, due to a lack of molecular methods and related infrastructure, authors were unable to do molecular typing of different MRSA strains and to work out their spread among the hospital environment. There may be other risk factors that need to be evaluated in further studies.
Declarations
Conflicts of Interest
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article
Authors Funding
The financial and infrastructure support was provided by the Dept. of Microbiology, I.G.I.M.S, Patna as per directions by the Institutional Ethical Committee. No external funding was utilized in this study.
Author’s Contributions
Dr. Suprakash Das reporting microbiologist and preparation of manuscript. Dr. Keshav Kumar Bimal collected and analyzed the data. Dr. Namrata Kumari assisted in paper writing and statistical analysis. Dr. Shailesh Kumar assisted in drafting the manuscript and critical revision.
References
- Tong, Steven YC, et al. "Staphylococcus aureus infections: Epidemiology, pathophysiology, clinical manifestations, and management." Clinical Microbiology Reviews, Vol. 28, No. 3, 2015, pp. 603-61.
- Grundmann, Hajo, et al. "Emergence and resurgence of meticillin-resistant Staphylococcus aureus as a public-health threat." The Lancet, Vol. 368, No. 9538, 2006, pp. 874-85.
- Chambers, Henry F., and Frank R. DeLeo. "Waves of resistance: Staphylococcus aureus in the antibiotic era." Nature Reviews Microbiology, Vol. 7, No. 9, 2009, pp. 629-41.
- Herold, Betsy C., et al. "Community-acquired methicillin-resistant Staphylococcus aureus in children with no identified predisposing risk." JAMA, Vol. 279, No. 8, 1998, pp. 593-98.
- Liu, Catherine, et al. "Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children." Clinical Infectious Diseases, Vol. 52, No. 3, 2011, pp. e18-55.
- Boucher, Helen, Loren G. Miller, and Raymund R. Razonable. "Serious infections caused by methicillin-resistant Staphylococcus aureus." Clinical Infectious Diseases, Vol. 51, No. Supplement_2, 2010, pp. S183-97.
- Wenzel, Richard P., et al. "Methicillin-resistant Staphylococcus aureus: Implications for the 1990s and effective control measures." The American Journal of Medicine, Vol. 91, No. 3, 1991, pp. S221-27.
- Rosenthal, Victor D., et al. "International Nosocomial Infection Control Consortium (INICC) report, data summary of 36 countries, for 2004-2009." American Journal of Infection Control, Vol. 40, No. 5, 2012, pp. 396-407.
- Stenehjem, Edward, and David Rimland. "MRSA nasal colonization burden and risk of MRSA infection." American Journal of Infection Control, Vol. 41, No. 5, 2013, pp. 405-10.
- Wagenvoort, J. H. T., et al. "MRSA from air-exhaust channels." The Lancet, Vol. 341, No. 8848, 1993, pp. 840-41.
- Layton, Marcelle C., et al. "An outbreak of mupirocin-resistant Staphylococcus aureus on a dermatology ward associated with an environmental reservoir." Infection Control and Hospital Epidemiology, Vol. 14, No. 7, 1993, pp. 369-75.
- Cosgrove, Sara E., et al. "Comparison of mortality associated with methicillin-resistant and methicillin-susceptible Staphylococcus aureus bacteremia: A meta-analysis." Clinical Infectious Diseases, Vol. 36, No. 1, 2003, pp. 53-59.
- Wooten, D. A., and L. G. Winston. "Risk factors for methicillin-resistant Staphylococcus aureus in patients with community-onset and hospital-onset pneumonia." Respiratory Medicine, Vol. 107, No. 8, 2013, pp. 1266-70.
- Sarma, J. B., and G. U. Ahmed. "Characterisation of methicillin resistant S. aureus strains and risk factors for acquisition in a teaching hospital in northeast India." Indian Journal of Medical Microbiology, Vol. 28, No. 2, 2010, p. 127.
- Wayne, P. A. "Clinical and laboratory standards institute: Performance standards for antimicrobial susceptibility testing: 21th informational supplement." CLSI document M100-S20, 2014.
- Mehta, A. A., et al. "A pilot programme of MRSA surveillance in India. (MRSA Surveillance Study Group)." Journal of Postgraduate Medicine, Vol. 42, No. 1, 1996, pp. 1-3.
- Verma, Sheetal, et al. "Growing problem of methicillin resistant staphylococci--Indian scenario." Indian Journal of Medical Sciences, Vol. 54, No. 12, 2000, pp. 535-40.
- Anupurba, S., et al. "Prevalence of methicillin resistant Staphylococcus aureus in a tertiary referral hospital in eastern Uttar Pradesh." Indian Journal of Medical Microbiology, Vol. 21, No. 1, 2003, pp. 49-51.
- Rajaduraipandi, K., et al. "Prevalence and antimicrobial susceptibility pattern of methicillin resistant Staphylococcus aureus: A multicentre study." Indian Journal of Medical Microbiology, Vol. 24, No. 1, 2006, p. 34.
- Tsering, Dechen C., Ranabir Pal, and Sumit Kar. "Methicillin-resistant Staphylococcus aureus: Prevalence and current susceptibility pattern in Sikkim." Journal of Global Infectious Diseases, Vol. 3, No. 1, 2011, pp. 9-13.
- Chen, C-J., and Y-C. Huang. "New epidemiology of Staphylococcus aureus infection in Asia." Clinical Microbiology and Infection, Vol. 20, No. 7, 2014, pp. 605-23.
- Eshwara, Vandana Kalwaje, et al. "Staphylococcus aureus bacteremia in an Indian tertiary care hospital: Observational study on clinical epidemiology, resistance characteristics, and carriage of the Panton-Valentine leukocidin gene." International Journal of Infectious Diseases, Vol. 17, No. 11, 2013, pp. e1051-55.
- Waness, Abdelkarim. "Revisiting methicillin-resistant Staphylococcus aureus infections." Journal of Global Infectious Diseases, Vol. 2, No. 1, 2010, pp. 49-56.
- Winkler, J., et al. "Nasal carriage of Staphylococcus aureus: Correlation with hormonal status in women." Journal of Infectious Diseases, Vol. 162, No. 6, 1990, pp. 1400-02.
- Humphreys, Hilary, Fidelma Fitzpatick, and Brian J. Harvey. "Gender differences in rates of carriage and bloodstream infection caused by methicillin-resistant Staphylococcus aureus: Are they real, do they matter and why?" Clinical Infectious Diseases, Vol. 61, No. 11, 2015, pp. 1708-14.
- Santosaningsih, Dewi, et al. "Risk factors for methicillin-resistant Staphylococcus aureus carriage among patients at admission to the surgical ward in a resource-limited hospital in Indonesia." The American journal of Tropical Medicine and Hygiene, Vol. 97, No. 5, 2017, pp. 1310-12.
- Arora, Shilpa, et al. "Prevalence of methicillin-resistant Staphylococcus aureus (MRSA) in a tertiary care hospital in Northern India." Journal of Laboratory Physicians, Vol. 2, No. 2, 2010, pp. 78-81.
- Jadin, Sarah A. "Describing the varying definitions for multidrug resistant Enterobacteriaceae." American Journal of Infection Control, Vol. 42, No. 6, 2014, p. S32.
- VanEperen, Alison S., and John Segreti. "Empirical therapy in methicillin-resistant Staphylococcus aureus infections: An up-to-date approach." Journal of Infection and Chemotherapy, Vol. 22, No. 6, 2016, pp. 351-59.
- Paul, Mical, et al. "Importance of appropriate empirical antibiotic therapy for methicillin-resistant Staphylococcus aureus bacteraemia." Journal of Antimicrobial Chemotherapy, Vol. 65, No. 12, 2010, pp. 2658-65.
- Harinstein, L., J. Schafer, and F. D'Amico. "Risk factors associated with the conversion of meticillin-resistant Staphylococcus aureus colonisation to healthcare-associated infection." Journal of Hospital Infection, Vol. 79, No. 3, 2011, pp. 194-97.
- Yao, Zhenjiang, et al. "Healthcare associated infections of methicillin-resistant Staphylococcus aureus: A case-control-control study." Plos One, Vol. 10, No. 10, 2015, p. e0140604.
- Yamakawa, Kazuma, et al. "Assessment of risk factors related to healthcare-associated methicillin-resistant Staphylococcus aureus infection at patient admission to an intensive care unit in Japan." BMC Infectious Diseases, Vol. 11, No. 1, 2011, pp. 1-7.
- Wu, Huang-Pin, et al. "Liver cirrhosis and diabetes mellitus are risk factors for Staphylococcus aureus infection in patients with healthcare-associated or hospital-acquired pneumonia." Pulmonary Medicine, Vol. 2016, 2016.
- Alvarez, Carlos, Jaime Labarca, and Mauro Salles. "Prevention strategies for methicillin-resistant Staphylococcus aureus (MRSA) in Latin America." Brazilian Journal of Infectious Diseases, Vol. 14, 2010, pp. 107-08.
- Gould, I. M., et al. "Costs of healthcare-associated methicillin-resistant Staphylococcus aureus and its control." Clinical Microbiology and Infection, Vol. 16, No. 12, 2010, pp. 1721-28.