Research Article - International Journal of Medical Research & Health Sciences ( 2022) Volume 11, Issue 12
To Asses CSF Glucose, Micro-Protein and CSF ADA Levels and Clinically Importance for Tuberculous Maningitis in Children
Afsha Mohi-UD-Din1, Nita Garg2, Mohammad Shahrukh1* and Kiran Bhatt32Department of Biochemistry, Autonomous State Medical College and Hospital, Firozabad, UP, India
3Department of Biochemistry, Swami Rama Himalayan University (SRHU), Dehradun, Uttrakhand, India
Mohammad Shahrukh, Department of Biochemistry, Shri Guru Ram Rai Institute of Medical and Health Science, Dehradun, Uttrakhand, India, Email: mohammadshahrukh.saifi@gmail.com
Received: 22-Dec-2021, Manuscript No. IJMRHS-21-50470; Editor assigned: 27-Dec-2021, Pre QC No. IJMRHS-21-50470(PQ); Reviewed: 10-Jan-2022, QC No. IJMRHS-21-50470; Revised: 30-Nov-2022, Manuscript No. IJMRHS-21-50470(R); Published: 28-Dec-2022
Abstract
Meningitis is inflammation of the meninges which affect the dura matter, piamatter and arachnoid membranes of the brain. The study was conducted on 79 subjects, out of 79 subjects, 59 patients with tuberculous meningitis and 20 patients with non tuberculous meningitis. In this study, assessed the levels of CSF glucose, micro protein and CSF ADA and knows the important role of these biochemical markers in tuberculous meningitis. In our study CSF glucose level was not significant, CSF micro protein level was significant and CSF ADA level was significant in cases as compared to control group.
Keywords
Tuberculous meningitis, CSF glucose, Micro protein, Brain
Introduction
Meningitis is inflammation of the meninges which comprise the dura matter, piamatter and arachnoid membranes. It may be caused by bacterial, fungal or viral infection, cerebral abscess, demyelinating disorders and malignant disease.
Tuberculous meningitis is also known as TB meningitis or tubercular meningitis. Tuberculous meningitis is mycobacterium tuberculosis infection of the meninges the system of membranes which envelop the central nervous system. Tuberculous Meningitis (TBM) is the most frequent form of Central Nervous System (CNS) tuberculosis. CNS disease accounts for only 5% of all cases of extra pulmonary tuberculosis and peak incidence is in children under 4 years of age.
Tuberculous Meningitis (TBM) continues to be the most common manifestation of neurotuberculosis in children. The classic presentation is with a subacutemeningitic illness, which can be difficult to distinguish from other causes of meningoencephalitis. Once the neurological symptoms of advanced disease are present (e.g., coma, seizures, raised intracranial pressure and hemiparesis), the diagnosis is apparent but the prognosis is poor. TBM is classified into three grades of severity according to the British medical research council TBM grade. Grade 1 TBM is defined as a Glasgow Coma Score (GCS) of 15 with no focal neurology; grade 2 TBM as a GCS of 15 with a focal neurological deficit, or a GCS of 11-14 and grade 3 TBM are defined as a GCS of ≤ 10. The importance of this classification system is that it enables stratification of patients and is useful to predict prognosis. Some children may present with ‘tuberculous encephalopathy’ with disseminated tuberculosis but without clinical or CSF evidence of meningitis [1].
In recent years, an elevated CSF Adenosine Deaminase (ADA) activity has been shown to be a promising test for the differentiation of patients of TBM. The results were less reliable in children. As a marker of cellular immunity, ADA activity is found to be elevated in those diseases in which there is a cell mediated immune response. Raised levels of enzyme have been found in tuberculouspleural, peritoneal and pericardial fluids and Cerebrospinal Fluid (CSF) of patients with TBM. In view of these observations ADA activity was estimated in CSF of children with tubeculous meningitis, partially treated meningitis, aseptic meningitis and pyogenic meningitis to assess its diagnostic value for TBM and to correlate with stages of TB and clinical parameters of CMI response to tuberculous infection [2].
Aim and objective
Aim: To assess the clinical significance of tuberculous meningitis in children.
Objective:
• To estimate CSF glucose and micro protein in tuberculosis meningitis patients.
• To estimate CSF ADA levels and its role in tuberculosis meningitis.
Materials and Methods
The study was conducted in the department of biochemistry, at Shri Mahant Indiresh hospital attached to the Shri Guru Ram Rai institute of medical and health science, Patel Nagar, Dehradun, Uttarakhand, India.
Source of data
Total patients-79 (including cases (59) and control (20)).
The samples were taken from the patients who visited at SMI hospital Dehradun. Subjects were recruited according to simple random sampling method that met the selection criteria. The persons of all the age groups from 1 month to 10 years were taken. Both the gender was included in the study [3-5].
Inclusion criteria:
• Patients with physical findings of tubercular meningitis in CSF.
• Patient who had tuberculosis or history of tuberculosis.
• Includes patients of both the genders and age group of 10 and below years.
Exclusion criteria:
• Patients below the age of 15 years.
• Those who are not willing to participate.
• CSF glucose was estimates by GOD/POD method (VITROS chemistry products GLU slides quantitatively measure Glucose (GLU) concentration in serum, plasma, urine and CSF using VITROS 250/350/950 and 5,1 FS chemistry systems and the VITROS 5600 integrated system.
• Micro protein was estimated by pyrogallol red method (For the quantitative determination of micro protein in urine and CSF).
• CSF ADA was estimated by MICROXPRESS ADA-MTB method (Colorimetric method).
Results
The evaluation of the data of subjects indicated that the enrolled subjects were distributed according to different trends. They were distributed according to age, gender, activity of ADA, glucose and micro protein levels in CSF test group (Tables 1-4 and Figures 1-4) [6-8].
| Age distribution | Cases | Control | ||
|---|---|---|---|---|
| No of patients | Percentage (%) | No of patients | Percentage (%) | |
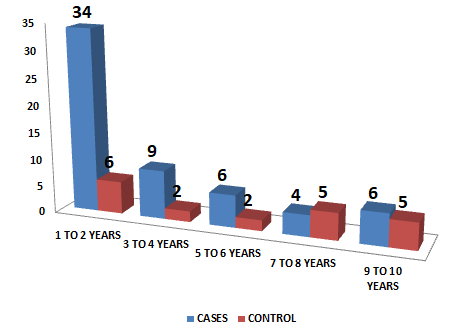
| 1-2 years | 34 | 57.62% | 6 | 30.00% |
| 3-4 years | 9 | 15.25% | 2 | 10.00% |
| 5-6 years | 6 | 10.16% | 2 | 10.00% |
| 7-8 years | 4 | 6.77% | 5 | 25.00% |
| 9-10 years | 6 | 10.16% | 5 | 25.00% |
| Gender distribution | Cases | Control | ||
|---|---|---|---|---|
| No of patients | Percentage (%) | No of patients | Percentage (%) | |
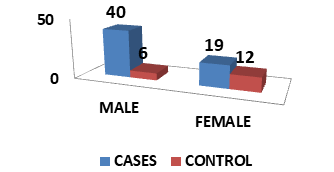
| Male | 40 | 67.8 | 8 | 40% |
| Female | 19 | 32.20% | 12 | 60% |
| Total | 59 | 100% | 20 | 100% |
| Age groups | ADA activity |
|---|---|
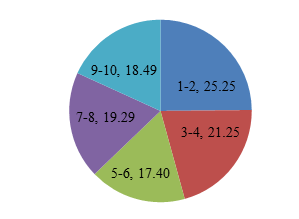
| 1-2 | 25.25 |
| 3-4 | 21.25 |
| 5-6 | 17.4 |
| 7-8 | 19.29 |
| 9-10 | 18.49 |
| Parameter | Cases mean ± SD | Control mean ± SD | P value | Significance |
|---|---|---|---|---|
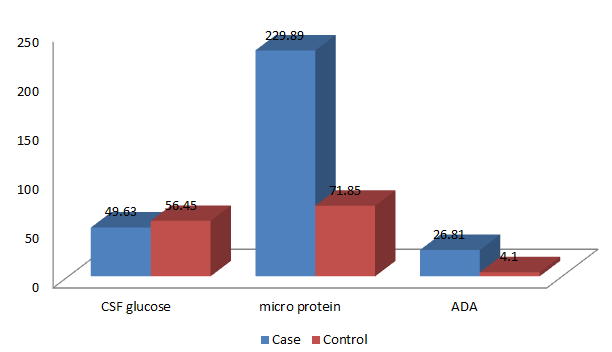
| CSF (glucose) | 49.63 ± 24.57 | 56.45 ± 30.25 | 0.31 | Not significant |
| µ protien | 229.89 ± 209.05 | 71.85 ± 61.88 | 0.001 | Significant |
| CSF ADA | 26.81 ± 21.29 | 4.13 ± 3.10 | 0.001 | Significant |
CSF glucose mean ± SD value was 49.63 ± 2 4.57, micro protein mean ± SD value was 229.89 ± 209.05 and CSF ADA mean ± SD value was 26.81 ± 21.29 in cases. CSF glucose mean ± SD value was 56.45 ± 30.25; micro protein mean ± SD value was 71.85 ± 61.88 and CSF ADA mean ± SD value was 4.13 ± 3.10 in normal subjects.
Discussion
The present study was planned to study the diagnostic utility of cerebral spinal fluid glucose, micro protein and CSF ADA in tuberculous meningitis. The study comprised of total 79 subjects, out of which the test group comprised of 59 patients i.e., group of people that had tubercular meningitis and admitted in the medicine and pulmonary medicine ward of Shri Mahant Indiresh hospital, Dehradun. The control group comprised of twenty subjects therefore the group of people that had not the tuberculous meningitis and were healthy population [9-12].
Age
According to age, subjects were distributed into five age groups 1-2 years, 3-4 years, 5-6 years, 7-8 years and 9-10 years. In the present study was observed that maximum number of tubercular meningitis patients were in third decade i.e., age group 1-2 years represented by 34 patients (57.62 %), 3-4 years represented by 9 patients (15.25%), 5-6 years represented by 6 patients (10.16%), 7-8 years represented by 4 patients (6.77 %) and 9-10 years represented by 6 patients (10.16%). Other age groups in control age group 1-2 years represented by 6 patients (30.0 %), 3-4 years represented by 2 patients (10.0%), 5-6 years represented by 2 patients (10.0%), 7-8 years represented by 5 patients (25.0 %) and 9-10 years represented by 5 patients (25.0 %).
In a study of A. Pan had total of 62 subjects, of them 43 were boys and 19 were girls. The mean age of children was 4.15 years in TBM. In the study of Bindu TH, et al., had 55 patients, out of fifty five patients 32 patients fulfilled the criteria were labeled as tubercular while other 23 patients were labeled as non-tubercular. The mean age of children is 4.36 years.
In the present study we have total 79 patients, out of 79 patients 59 cases and 20 control groups, the mean age is 3.8 years.
CSF glucose
In our study we observed the value of CSF glucose is 49.63 mg/dl ± 24.57 mg/dl in study group and 56.45 mg/dl ± 30.25 mg/dl in control group. The CSF glucose level is not significant in cases as compared to control group (Table 5).
| Mean value of CSF glucose | Significance |
|---|---|
| 37.9 ± 18.3 | Not significant |
| 45.92 ± 1.91 | Not significant |
| 33.656 ± 20.185 | Not significant |
| 49.63 ± 24.57 | Not significant |
CSF ADA
In our study we observed the value of CSF ADA is 26.81 IU/L ± 21.29 05 IU/L in study group and 4.13 IU/L ± 3.10 IU/L in control group (Table 6).
| Mean value of CSF ADA | Significance |
|---|---|
| 6.43 ± 1.93 | Significant |
| 9.4 ± 5.4 | Significant |
| 18.22 ± 3.35 | Significant |
| 11.38 ± 0.77 | Significant |
| 13.3250 ± 4.49344 | Significant |
| 26.81 ± 21.29 | Significant |
The mean ADA levels in CSF in cases with tuberculosis meningitis have been reported to be higher as compared to control group.
Conclusion
As few studies have been conducted to evaluate the role of CSF ADA levels for the diagnosis of TBM, we tried to estimate ADA levels in CSF in TBM and to find out its role as a sensitive, accurate, rapid, and affordable diagnostic tool that will work in resource limited settings in confirming the tubercular etiology in cases of meningitis. (Adenosine deaminase levels in CSF of tuberculous meningitis patients.
In tubercular meningitis we found:
• CSF glucose level was lower and not significant in cases as compared to control group.
• CSF micro protein level was higher and significant in cases as compared to control group.
• In the present study, CSF ADA level was higher and significant in cases as compared to control group.
References
- Mason P, et al. Handbook of pharmacy healthcare: Diseases and patient advice. 2nd edition, Pharma Press, United States, 2002.
[Googlescholar] [Indexed]
- Torok ME, et al. Tuberculous meningitis: Advances in diagnosis and treatment. British Med Bulletin, Vol. 113, No. 1, 2015, pp. 117-131.
[Crossref] [Googlescholar] [Indexed]
- Upadhyay GC, et al. Tuberculous meningitis in children: A clinicolaboratory correlation of CSF findings for early diagnosis. Indian J Pediatr, Vol. 51, No. 6, 1984, pp. 633-666.
[Crossref] [Googlescholar] [Indexed]
- Alarcon F, et al. Movement disorders in 30 patients with tuberculous meningitis. Movement Disorders: Official J Movement Disorder Society, Vol. 15, No. 3, 2000, pp. 561-569.
[Crossref] [Googlescholar] [Indexed]
- Banales JL, et al. Adenosine deaminase in the diagnosis of tuberculous pleural effusions: A report of 218 patients and review of the literature. Chest, Vol. 99, No. 2, 1991, pp. 355-357.
[Crossref] [Googlescholar] [Indexed]
- Pan A, et al. Evaluation of cerebrospinal fluid adenosine deaminase activity as a complimentary tool in the early diagnosis of tuberculous meningitis in children. J College Med Sci Nepal, Vol. 12, No. 8, 2012, pp. 17-23.
[Crossref] [Googlescholar] [Indexed]
- Wiczkowski A, et al. Acid-base balance and electrolytes in cerebrospinal fluid of patients with bacterial and lymphocytic meningitis. Przeglad lekarski, Vol. 52, No. 10, 1995, pp. 509-512. [Crossref] [Googlescholar] [Indexed]
- Bindu TH, et al. Role of cerebrospinal fluid adenosine deaminase activity in the diagnosis of tuberculous meningitis in children. Int J Contemp Pediatr, Vol. 4, 2017, pp. 411.
[Crossref] [Googlescholar] [Indexed]
- Mishra OP, et al. Cerebrospinal fluid adenosine deaminase activity for the diagnosis of tuberculous meningitis in children. J Tropical Pedia, Vol. 42, No. 3, 1996, pp. 129-132.
[Crossref] [Googlescholar] [Indexed]
- Siddiqui B, et al. Comparitive biochemical profile of cerebrospinal fluid and serum in the diagnosis of pyogenic meningitis. Pharma Innov, Vol. 5, No. 2, 2016, pp. 1.
[Googlescholar] [Indexed]
- P. Rajendr, et al. Adenosine deaminase activity in cerebro-spinal fluid for diagnosis of tuberculous meningitis. Ind J Tub, Vol. 38, 1991, pp. 99. [Crossref]
[Googlescholar] [Indexed]
- Rana SV, et al. Adenosine deaminase levels in cerebrospinal fluid as a diagnostic test for tuberculous meningitis in children. Indian J Clin Biochem. Vol. 19, No. 2, 2004, pp. 5.
[Crossref] [Googlescholar] [Indexed]