Editorial - International Journal of Medical Research & Health Sciences ( 2023) Volume 12, Issue 4
Adverse Effects of Proton Pump Inhibitors use Among South Asian Population Systematic Review
Samprith Ala1*, Mogan Saruveish2, Cook Daniel Elkin1, Junaid Imran3, Bilal Abu Tarboush4 and Vinoshini Muthusamy52Faculty of Medicine and Health Sciences, Universiti Malaysia Sarawak, Malaysia
3Medical College Peshawar, Pakistan
4Faculty of Medicine, Universite Djillali Liabes-Sidi Bel Abbes, Algeria
5Kementerian Kesihatan, Malaysia
Samprith Ala, Avalon University School of Medicine,Curacao, Iceland, Email: samprithala@gmail.com
Received: 29-Mar-2023, Manuscript No. ijmrhs-23-93542; Editor assigned: 01-Apr-2023, Pre QC No. ijmrhs-23-93542(PQ); Reviewed: 05-Apr-2023, QC No. ijmrhs-23-93542(Q); Revised: 12-Apr-2023, Manuscript No. ijmrhs-23-93542(R); Published: 30-Apr-2023
Abstract
Background: This systematic review aims to identify the adverse effects of Proton Pump Inhibitors (PPI) among the South Asian population.
Methods: A literature review was conducted using PubMed and EBSCO databases following the PRISMA guidelines. The search was performed between December 2022 and February 2023 using the Population, Intervention, Control, and Outcome (PICO) framework to develop a search syntax. Inclusion and exclusion criteria were established, and the search terms were used to identify relevant articles.
Results: The search identified studies conducted in India, Pakistan, Bangladesh, and Indonesia. The adverse effects of PPI use in this population included acute kidney injury, an increased risk of osteoporosis and fracture, tubulointerstitial nephritis, QT prolongation and an increased risk of torsade's de pointes, diabetic nephropathy, an increased risk of thrombosis when combined with clopidogrel, constipation, and vitamin B12 deficiency.
Conclusion: The use of PPIs among the South Asian population is associated with several adverse effects, including an increased risk of serious health conditions. Clinicians should exercise caution when prescribing PPIs to this population and weigh the benefits against the potential risks. Further research is needed to better understand the adverse effects of PPI use among South Asians.
Keywords
South Asian, Proton Pump Inhibitors, Adverse effects, Ppi use, Side effects
Introduction
Proton Pump Inhibitors (PPIs) irreversibly inhibit the H+ K+ ATPase. A range of medical conditions is treated with Proton pump inhibitors (PPIs), including gastric ulcers, gastroesophageal reflux disease, dyspepsia, Zollinger Ellison syndrome, bleeding peptic ulcers, Helicobacter pylori eradication, esophagitis, and prevention of gastric ulcers induced by nonsteroidal anti-inflammatory drugs [1]. PPIs are increasingly being used due to their superior effectiveness compared to other drugs, and their worldwide annual sales have exceeded $25 billion. Although PPIs are usually deemed safe, their usage may result in several consequences, such as acute kidney injury, Tubulo-interstitial nephritis, diarrhea, vitamin B12 deficiency, osteoporosis, diabetic nephropathy, and hyperprolactinemia [2]. PPIs also raise the likelihood of developing Clostridium difficile infections, experiencing cardiovascular events, and having prolonged QT intervals [3-12]. The elderly population is at risk of experiencing increased morbidity, adverse drug reactions, hospitalization, and even death as a result of overuse of PPIs, which can be attributed to their susceptibility to multiple comorbidities and the concurrent use of multiple medications. In patients with COVID-19, taking PPIs also elevated the chances of experiencing negative combined outcomes [13,14].
The objective of our study is to investigate and analyze the possible adverse effects of Proton pump inhibitors (PPIs) in the context of South Asian populations, intending to provide a comprehensive understanding of their safety profile in this specific demographic.
Materials and Methods
The literature review followed the PRISMA guidelines [15]. The search for relevant literature was conducted between December 1 2022 and February 01 2023. Before conducting the review, a research protocol was established and agreed upon, although it was not registered or published.
Search Strategy
The search for relevant articles was conducted using two databases, PubMed and EBSCO. The research team utilized the Population, Intervention, Control, and Outcome (PICO) framework to develop a search syntax. By applying the PICO concepts in the search query, the currently unanswered questions in community settings were highlighted [16]. The team summarized the use of the PICO framework in selecting keywords for the review in Table 1.
| PICO | CONCEPTS |
|---|---|
| P- POPULATION | South Asian |
| I-INTERVENTION | PPI Use |
| C- CONTROL | Not Applicable |
| O- OUTCOME | Adverse Effects |
Table 1 provided the PICO concepts, which were used to generate the keywords and MESH terms listed below. These search terms were used to identify relevant articles for the systematic review in PubMed. The PubMed search involved the use of both free keywords and MESH terms, whereas the search in EBSCO only utilized free keywords.
Free Terms
• South Asia or India or Nepal or Bangladesh or Pakistan or Indian subcontinent or Sri Lanka or Bhutan or Maldives or Afghanistan.
• Proton pump inhibitor or ppi or pantoprazole or omeprazole or lansoprazole rabeprazole or esomeprazole or dexlansoprazole.
• Adverse effects or side effects or complications or unfavorable or disadvantageous.
Mesh Terms
• “Asia, Southern/epidemiology”(Mesh).
• Proton Pump Inhibitors"(Mesh)or "Proton Pump Inhibitors" (Pharmacological Action) or "Proton Pumps"(Mesh).
• "Adverse effects" (Subheading) or "Long Term Adverse Effects"(Mesh).
Inclusion and Exclusion Criteria
the inclusion and exclusion criteria for relevant articles, focus on studies written in English and involve populations from South Asia who use any form of PPI but exclude studies that examine H2 blockers use or are conducted in countries outside of South Asia.
Inclusion Criteria
• Papers Written in English.
• Population from South Asia.
• Any form of PPI use.
Exclusion Criteria
• Country of interest not in South Asia.
• Articles in any language other than English.
• H2 blockers use.
Selection Criteria
The screening process was divided into three phases. In the first phase, the titles and abstracts of all publications identified in the search were examined, and any discrepancies were resolved to determine whether a publication should be included. The second phase involved reviewing the full texts of relevant publications from the first phase, which required contacting some authors to obtain the complete articles as they were not readily available. Some authors responded to the request by sending their publications. In the third and final phase, the reference lists of the papers selected in the second phase were reviewed. The same inclusion and exclusion criteria were applied during all three screening phases.
Results
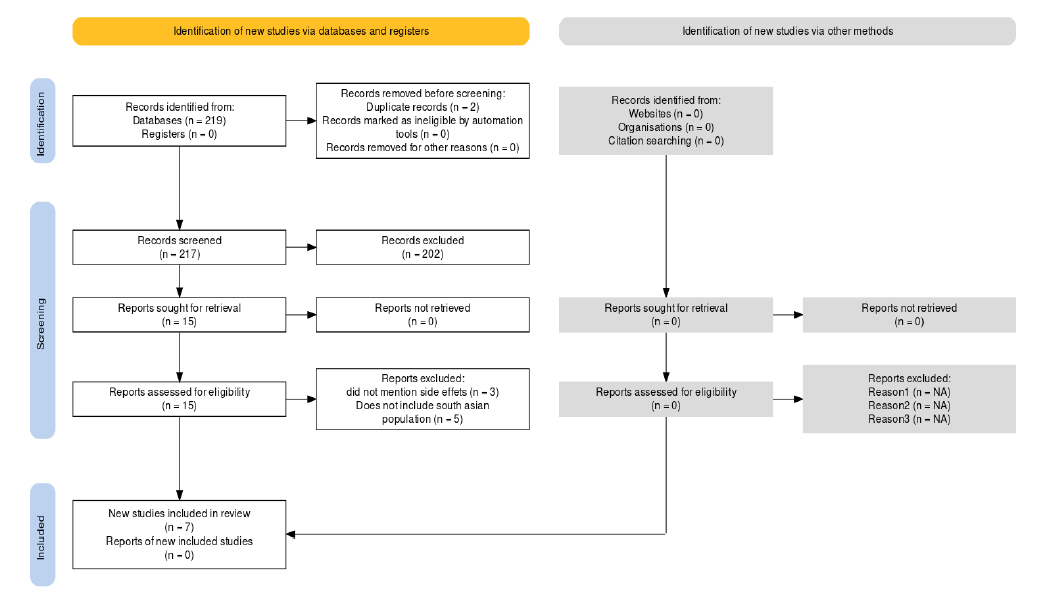
The paragraph outlines the method of filtering publications by applying certain criteria for inclusion and exclusion. Initially, a total of 219 publications were collected and evaluated against the said criteria. Two articles were removed as duplicates, leaving 217 publications to be screened. Among these, 202 publications were excluded for not meeting the inclusion criteria, and 15 articles were considered for full-text assessment. Ultimately, seven publications were excluded, and eight studies were deemed eligible for inclusion in the study. A visual representation of the selection process is presented in Figure 1, while Appendix A provides a list of all the articles included in the study.
General Characteristics of the Publications
Table 2 presents a general description of the included publications. Our review included all types of studies, namely interventional and non-interventional studies in South Asian countries including India, Pakistan, and Bangladesh.
| Classification category | Subcategories | N (%)* | For reference index, see Appendix A |
|---|---|---|---|
| Country of research | India | 42.825 | 1,2,4 |
| Pakistan | 42.825 | 3,6,7 | |
| Bangladesh | 14.285 | 5 | |
| Year of publication | 2022 | 42.825 | 2,3,5 |
| 2020 | 14.285 | 6 | |
| 2019 | 14.285 | 7 | |
| 2017 | 14.285 | 1 | |
| 2013 | 14.285 | 4 | |
*Percentages show the share of the total publications reviewed |
|||
Summary of Evidence
According to evidence, there are numerous side effects associated with using proton pump inhibitors. Some of them were acute kidney injury and increased risk of osteoporosis. These side effects are summarized in Table 3.
| Country | Adverse Effects |
|---|---|
| India, Pakistan, Bangladesh | Acute Kidney Injury, increase risk of osteoporosis and fracture, |
| Tubulointerstitial nephritis, QT prolongation and increase risk of | |
| Torsade's de pointes, diabetic nephropathy, increase risk of | |
| thrombosis when combined proton pump inhibitors with clopidogrel, | |
| vitamin B12 deficiency |
Discussion
Based on the systematic review conducted, it was found that there are several adverse effects associated with the use of Proton Pump Inhibitors (PPIs) in South Asian countries, including India, Pakistan, Bangladesh, and Indonesia. These adverse effects include acute kidney injury, increased risk of osteoporosis and fractures, tubulointerstitial nephritis, QT prolongation, and increased risk of Torsade's de pointes, diabetic nephropathy and increased risk of thrombosis when combined with clopidogrel [3,8,10,17,18]. There is also evidence that primarily focuses on shortterm PPI therapy and suggests that there may be a potential link between PPIs and the development of sexual and reproductive disorders, such as elevated serum prolactin levels. However, the available literature is insufficient to establish a definite relationship between PPI use and prolactin levels [3]. Compared to other studies, the incidence of constipation among patients who took proton pump inhibitors in this study conducted at Gatot Soebroto Hospital was relatively high [6].
Patients with diabetes mellitus are at a higher risk of developing peptic ulcer disease and may require treatment with Proton Pump Inhibitors (PPIs) to manage this condition. While PPIs can provide clinical benefits for peptic ulcer disease, there is evidence to suggest that their use may contribute to the development of diabetic nephropathy. As such, caution should be exercised when prescribing PPIs to patients who have other risk factors for diabetic nephropathy [10]. When prescribing Proton Pump Inhibitors (PPIs) to patients receiving dual antiplatelet therapy, it is important to conduct a comprehensive risk-benefit assessment. The potential drug interaction between PPIs and antiplatelet therapy needs further clarification through randomized controlled trials. Several studies have demonstrated that administering proton pump inhibitors other than pantoprazole in combination with Clopidogrel can lead to a loss of its beneficial effects and an increased risk of re-infarction. This may occur due to competitive interference in the P450 pathway, which impedes the biotransformation of Clopidogrel into its active drug form necessary for effective platelet inhibition. Omeprazole, in particular, has been shown to inhibit the antiplatelet activity of Clopidogrel by inhibiting CYP2C19. Therefore, caution should be exercised when considering the use of PPIs in patients receiving dual antiplatelet therapy, and healthcare professionals should be aware of the potential risks and benefits. Further research is needed to clarify the extent of this drug interaction and to identify the most appropriate treatment strategies for patients requiring both PPIs and antiplatelet therapy [8].
It should be noted that the studies included in this systematic review varied in terms of their design, sample size, and methods of data analysis. This may have contributed to some degree of heterogeneity in the findings. Nonetheless, the results of this review provide valuable insights into the adverse effects of PPIs in South Asian populations and highlight the need for further research in this area.
Overall, this systematic review adds to the growing body of literature on the adverse effects of PPIs and underscores the importance of careful consideration and monitoring of patients who are prescribed these medications. It is hoped that the findings of this review will inform clinical practice and contribute to improved patient outcomes in South Asian populations.
Conclusion
In conclusion, this systematic review highlights the adverse effects associated with the use of Proton Pump Inhibitors (PPIs) in South Asian populations. These adverse effects include acute kidney injury, increased risk of osteoporosis and fractures, tubulointerstitial nephritis, QT prolongation and increased risk of Torsade's de pointes, diabetic nephropathy, and increased risk of thrombosis when combined with clopidogrel.
Furthermore, patients, especially those with pre-existing conditions that raise their risk of experiencing negative side effects, should be prescribed PPIs with caution. Healthcare professionals should also keep a close eye on patients for any indications of negative side effects, especially those connected to prolonged PPI use. Although the designs and analytical techniques used by the studies included in this review varied, the conclusions offer important new information about the dangers of PPI use in South Asian populations. The extent of drug interactions needs to be clarified, and the best treatment plans for patients who need both PPIs and antiplatelet therapy need to be determined.
Declarations
Conflict of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research did not receive any specific grant or funding from agencies, the public, commercial, or not-for-profit sectors.
Ethics Approval
Since this analysis was a systematic review, no human ethics approval was required.
Limitations
This study only reviewed publications written or translated into English sourced from two databases. The review also excluded grey data and studies published at the time of review.
References
- Fossmark, Reidar, Tom C. Martinsen, and Helge L. Waldum. "Adverse effects of proton pump inhibitors—evidence and plausibility." International journal of molecular sciences, Vol. 20, No. 20, 2019, p. 5203.
Google Scholar Crossref - Mitra, Shuvo, et al. "A survey on the incidence of common musculoskeletal side effects among the patients taking long-term anti-ulcerant therapies in Bangladesh." Toxicology Reports, Vol. 9, 2022, pp. 1796-805.
Google Scholar Crossref - Ashfaq, Muhammad, Muhammad Z. Haroon, and Yasser M. Alkahraman. "Proton pump inhibitors therapy and risk of hyperprolactinemia with associated sexual disorders." Endocrine Regulations, Vol. 56, No. 2, 2022, pp. 134-47.
Google Scholar Crossref - Bhatia, Shobna, et al. "An Expert Review and Recommendations on the Rational Use of Proton Pump Inhibitors: Indian Perspective." The Journal of the Association of Physicians of India, Vol. 67, No. 4, 2019, pp. 88-96.
Google Scholar - Biswas, Mohitosh, et al. "Risk of major adverse cardiovascular events for concomitant use of clopidogrel and proton pump inhibitors in patients inheriting CYP2C19 loss-of-function alleles: meta-analysis." International journal of clinical pharmacy, Vol. 43, 2021, pp. 1360-69.
Google Scholar Crossref - Hanif, Inas F., et al. "Gastrointestinal Side Effects of Proton Pump Inhibitors on Inpatients at Gatot Soebroto Hospital." Journal of Young Pharmacists, Vol. 9, No. 1, 2017, p. S13.
Google Scholar Crossref - Ingle, Meghraj, et al. "Prevalence and clinical course of Clostridium difficile infection in a tertiary-care hospital: a retrospective analysis." Indian Journal of Gastroenterology, Vol. 30, 2011, pp. 89-93.
Google Scholar Crossref - Kashyap, Mandavi, et al. "Drug-drug interactions and their predictors: Results from Indian elderly inpatients." Pharmacy practice, Vol. 11, No. 4, 2013, p. 191.
Google Scholar Crossref - Khan, Qasim, et al. "Prevalence of the risk factors for QT prolongation and associated drug–drug interactions in a cohort of medical inpatients." Journal of the Formosan Medical Association, Vol. 118, No. 1, 2019, pp. 109-15.
Google Scholar Crossref - Raj, Jeffrey Pradeep, et al. "Diabetic nephropathy and proton pump inhibitors–Pilot case-control study." Indian Journal of Nephrology, Vol. 32, No. 2, 2022, p. 127.
Google Scholar Crossref - Shehryar, Muhammad, et al. "Efficacy, safety and cost-effectiveness of vonoprazan vs Proton Pump Inhibitors in reflux disorders and H. pylori eradication: A literature review." Annals of Medicine and Surgery, 2022.
Google Scholar Crossref - Singh, Amandeep, et al. "Association between anti-acid therapies and advanced fibrosis in type 2 diabetics with biopsy-proven non-alcoholic fatty liver disease." Indian Journal of Gastroenterology, Vol. 39, 2020, pp. 591-98.
Google Scholar Crossref - Lavu, Alekhya, et al. "A Cross-Sectional Study on Single-Day Use of Proton Pump Inhibitors in Tertiary Care Hospitals of South India." Hospital pharmacy, Vol. 56, No. 2, 2021, p. 109.
Google Scholar Crossref - Fatima, Kaneez, et al. "The use of proton pump inhibitors and COVID-19: A systematic review and meta-analysis." Tropical Medicine and Infectious Disease, Vol. 7, No. 3, 2022, p. 37.
Google Scholar Crossref - Page, Matthew J., et al. "The PRISMA 2020 statement: an updated guideline for reporting systematic reviews." International journal of surgery, Vol. 88, 2021.
Google Scholar Crossref - Aslam, Sadaf, and Patricia Emmanuel. "Formulating a researchable question: A critical step for facilitating good clinical research." Indian journal of sexually transmitted diseases and AIDS, Vol. 31, No. 1, 2010, p. 47.
Google Scholar Crossref - Avinash, A., et al. "A retrospective study to assess the effect of proton pump inhibitors on renal profile in a south indian hospital." Journal of Clinical and Diagnostic Research: JCDR, Vol. 11, No. 4, 2017, p. FC09.
Google Scholar Crossref - Qureshi, Ruqaya, et al. "Frequency and etiology of tubulo-interstitial nephritis in an adult renal biopsies in a tertiary renal care hospital: a single-center study." Saudi Journal of Kidney Diseases and Transplantation, Vol. 31, No. 2, 2020, pp. 335-41.
Google Scholar Crossref
Appendix
A List of articles selected for review
| Publication number | Title | Year | Country |
|---|---|---|---|
| 1 | A Retrospective Study to Assess the Effect of Proton Pump Inhibitors on Renal Profile in a South Indian Hospital | 2017 | India |
| 2 | Diabetic Nephropathy and Proton Pump Inhibitors - Pilot Case-Control Study | 2022 | India |
| 3 | Proton pump inhibitors therapy and risk of hyperprolactinemia with associated sexual disorders | 2022 | Pakistan |
| 4 | Drug-drug interactions and their predictors: Results from Indian elderly inpatients | 2013 | India |
| 5 | A survey on the incidence of common musculoskeletal side effects among patients taking long-term anti-ulcerate therapies in Bangladesh | 2022 | Bangladesh |
| 6 | Frequency and etiology of tubulointerstitial nephritis in an adult renal biopsy in a tertiary renal care hospital: A single-center study | 2020 | Pakistan |
| 7 | Prevalence of the risk factors for QT prolongation and associated drug-drug interactions in a cohort of medical inpatients | 2019 | Pakistan |