Case Report - International Journal of Medical Research & Health Sciences ( 2023) Volume 12, Issue 8
Pantoprazole Induced Black Hairy Tongue: A Case Report
Divya G Krishnan1*, Shaikh Ubedulla Shaikh Iqbal Daud1, AnukeshVasuKeloth2 and Neethu P12Department of Surgery, KMCT Medical College, Kerala University of Health Sciences, Kerala, India
Divya G Krishnan, Department of Pharmacology, KMCT Medical College, Kerala University of Health Sciences, Kerala, India, Email: dgkindia@gmail.com
Received: 06-Jul-2023, Manuscript No. ijmrhs-23-105169; Editor assigned: 08-Jul-2023, Pre QC No. ijmrhs-23-105169(PQ); Reviewed: 25-Jul-2023, QC No. ijmrhs-23-105169(Q); Revised: 02-Aug-2023, Manuscript No. ijmrhs-23-105169(R); Published: 30-Aug-2023
Abstract
Introduction: Black Hairy Tongue (BHT) is characterized by abnormally hypertrophied and elongated filiform papillae, resulting in a blackish discoloration on the dorsal surface of the tongue. BHT has been reported as an adverse drug reaction to various categories of drugs. Nevertheless, cases of Pantoprazole-induced BHT have been rarely reported. Case Description: We present a case involving a 42-year-old female who was admitted to the Surgery ward due to postoperative wound infection. She had been prescribed oral Clarithromycin and Pantoprazole. On the third day of initiating these medications, she reported experiencing a blackish discoloration on the dorsal surface of her tongue along with a change in her sense of taste. Clinical and microscopic examinations led to a diagnosis of Black Hairy Tongue (BHT). There was a suspicion that BHT might be an adverse drug reaction specifically to Pantoprazole. Consequently, the administration of Pantoprazole was ceased, while Clarithromycin continued for the full prescribed course. The patient received guidance to scrape her tongue thrice daily, maintain proper hydration, and adhere to good oral hygiene practices. By the third day following the discontinuation of Pantoprazole, the black discoloration and taste alterations had completely resolved. Causality assessment using the WHO-UMC (World Health Organization - Uppsala Monitoring Centre) scale indicated a 'probable' association between the adverse drug reaction and Pantoprazole. Conclusion: Physicians need to be vigilant about the potential occurrence of Black Hairy Tongue (BHT) in patients using Pantoprazole, and they should recognize that discontinuing the medication, coupled with mechanical debridement and maintaining proper oral hygiene, leads to complete resolution of the condition.
Keywords
Black Hairy Tongue (BHT), Pantoprazole, Adverse drug reaction, Surgical wound infection
Introduction
Black Hairy Tongue (BHT) is a painless and benign condition characterized by an unusual enlargement of the filiform papillae on the upper surface of the tongue [1]. It is also referred to as keratomycosis linguae, melanotrichia, lingua villosa nigra, and hyperkeratosis of the tongue [2]. BHT is found in approximately 0.4% of the South Asian population [3]. Although BHT frequently exhibits a black or brown discoloration, instances of yellow, green, and blue discolorations have also been documented [4]. Generally, BHT lacks symptoms, yet some patients may encounter changes in their sense of taste, unpleasant odors, tongue itching, or a burning sensation [5]. The precise origins and mechanisms responsible for BHT remain incompletely understood. Factors such as smoking, the consumption of heavily pigmented beverages like black tea or coffee, inadequate oral hygiene, as well as conditions such as HIV infection, malignancy, trigeminal neuralgia, and the use of specific medications including antipsychotics, methyldopa, antidepressants, anticancer drugs, and antibiotics have all been linked to the development of BHT [6,7]. The purpose of this article is to present a case of Pantoprazole induced BHT, which has been rarely reported till date.
Case Report
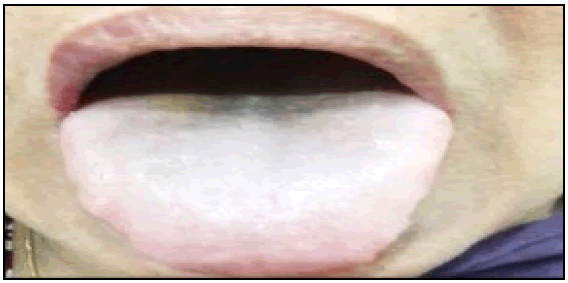
A 42-year-old female presented at the surgery outpatient department, complaining of pus discharge from her hernia repair wound. Diagnosis revealed a postoperative wound infection, leading to her admission to the surgery ward for treatment. Following the collection of pus from the wound for culture and sensitivity testing, the patient was initiated on empirical therapy. This included oral Clarithromycin 500 mg, to be taken twice daily, and oral Pantoprazole 40 mg, prescribed once daily as a preventive measure against drug-induced gastritis. By the third day after commencing the prescribed medications, the patient observed a blackish discoloration on the posterior section of the dorsal surface of her tongue (Figure 1). Additionally, she reported a change in her sense of taste. Notably, she did not experience pain, itching, a burning sensation, halitosis, or tongue bleeding. The patient denied any history of smoking, tobacco usage, or other habits detrimental to oral health. Consumption of coloured foods was also not a part of her history. Physical examination unveiled a blackish discoloration spanning the posterior one-third of the dorsal tongue surface. The patient maintained satisfactory oral hygiene, and her routine blood tests returned within normal ranges. Histopathological analysis of tongue scrapings indicated hypertrophied and keratinized filiform papillae. Upon careful evaluation, a tentative diagnosis of Black Hairy Tongue (BHT) arising as an adverse drug reaction to the administered medications was established. This diagnosis was arrived at following the exclusion of other potential causes of BHT. Given the suspicion that Pantoprazole was the triggering factor for BHT, the attending physician recommended discontinuing its use. Given that the surgical wound's pus culture sensitivity demonstrated susceptibility to Clarithromycin, the latter treatment was continued. The patient was instructed to gently scrape her tongue using a soft toothbrush three times a day, ensure adequate hydration, and uphold proper oral hygiene practices. Upon cessation of Pantoprazole, a noticeable improvement in the blackish discoloration and alteration in taste sensation was observed. By the third day after discontinuing Pantoprazole, the tongue regained its normal texture along with restored taste sensation. Pantoprazole was not reintroduced. The patient successfully completed the ten-day Clarithromycin course without further incidence of BHT. In this case WHO-UMC causality assessment scale showed a ‘probable’ association of the adverse drug reaction with Pantoprazole (Table 1).
| Causality term
|
Assessment criteria
|
|---|---|
| Certain
|
|
|
|
|
|
|
|
|
|
| Probable/Likely
|
|
|
|
|
|
|
|
| Possible
|
|
|
|
|
|
| Unlikely
|
|
|
|
| Conditional/ Unclassified
|
|
|
|
| Un-assessable/ Unclassifiable
|
|
|
|
|
Discussion
The first case of BHT was documented in 1557 by Dr. Amatus Lusitanus, who described the condition as selfrenewing of the tongue [1]. Pathophysiology of black hairy tongue has not been fully elucidated. It is thought to arise from defective desquamation of the dorsal surface of the tongue, which prevents normal debridement, leading to accumulation of keratinized layers and a hair-like appearance. Normally less than 1 mm in length but the elongated papillae can reach a length of 12 mm to 18 mm and width of 2 mm. These then secondarily collect fungi, bacteria, and debris [8].
Drug induced BHT have been frequently associated with the use of psychotropic drugs, anticancer drugs and antibiotics. Pantoprazole is a widely used Proton Pump Inhibitor (PPI) for the treatment of gastroesophageal reflux disease and prophylaxis for drug-induced gastritis. BHT has not been mentioned as a possible adverse reaction of PPI in the tertiary literature but there are isolated case reports of PPI induced BHT.
In 2009, 6 cases and 87 cases of PPI-induced BHT were reported in the database of the Netherlands Pharmacovigilance Centre Lab and WHO database respectively. Of the 6 cases from the Netherlands database, 5 cases were due to Omeprazole and 1 case was due to Pantoprazole. Of the 87 cases in the WHO database, 40 cases were due to Omeprazole, 22 cases due to Lansoprazole, 12 cases due to esomeprazole, 8 cases due to Pantoprazole and 5 cases due to Rabeprazole [9]. Hence, these reports show that BHT due to Pantoprazole is less frequently encountered as compared to other PPIs. Aryal et al reported a case of Lansoprazole induced BHT in 2017 [6].
PPI induced BHT is possibly due to alteration of the pH of gastric acid in the stomach and saliva and also a decrease in saliva production which may lead to defective desquamation of the dorsal surface of the tongue thereby accumulating a keratinized layer and allowing the growth of candida species and porphyrin producing chromogenic organisms in the oral flora giving the characteristic colour. Accumulation of keratinized layer causes hypertrophy and elongation of filiform papillae [10,11]. Another possible explanation is the use of red, black and yellow ferric oxide in capsules and tablets as a colorant which accumulates as debris with overgrowth of bacteria and yeasts which lead to synthesis of porphyrin producing chromogenic organisms resulting in BHT [12].
BHT presents with yellow to black tongue discoloration which may be associated with a burning sensation and taste alteration. Our patient presented with blackish discoloration and taste alteration which was similar to 3 cases in the Netherlands report. Time of onset of BHT with PPI has been shown to vary from 2 days to 2 years. Similarly resolution of BHT after stopping the offending medication is also variable. This could indicate that two different mechanisms for PPI induced tongue discoloration exist: altered microbial flora and discoloration by the colorant used in the capsule or tablet. In our case the onset and resolution of BHT were within 3 days of starting and 4 days of discontinuing Pantoprazole respectively which is similar to 2 cases due to Omeprazole in the Netherlands report. Use of antimicrobials may predispose to BHT induced by PPI. Among the PPI induced BHT cases in the 2009 WHO database, 24 cases were associated with concomitant therapy with antibiotics. Local and systemic antibiotics significantly alter oral flora, thus potentially predisposing the patient to develop BHT. This alteration of oral flora predispose to trapping of foreign material and stimulate local microbial overgrowth that lead to colour change with delayed desquamation of cells in central column of filiform papillae and marked retention of secondary papillary cells that express hair type keratins [13]. In our case, the use of Clarithromycin could have served as a predisposing factor but it cannot be positively concluded as the patient resolved after stopping Pantoprazole alone while Clarithromycin was given for the full course.
BHT can be successfully managed by stopping the offending agent, mechanical debridement like tongue scraping, maintaining good oral hygiene and adequate hydration [14]. In our case also the BHT resolved completely with these measures and no drug therapy was needed to treat it. Our report clearly indicates that BHT is reversible and unlikely to be harmful.
Conclusion
Pantoprazole induced BHT is a benign condition and can be successfully managed by stopping the medication, good oral hygiene and hydration. Physicians should be aware of the possibility of BHT with the use of Pantoprazole especially when administered with antibiotics. Documentation and reporting of such cases should be encouraged in order to have a more evidence based database on similar adverse drug reactions.
Acknowledgment
The authors express their gratitude to the Department of Surgery for furnishing valuable information pertaining to the patient under study.
Declarations
Ethical Considerations
All related research's ethical principles are considered in this article.
Conflict of Interest
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
References
- Schlager, Emma, et al. "Black hairy tongue: predisposing factors, diagnosis, and treatment." American journal of clinical dermatology, Vol. 18, 2017, pp. 563-9.
Google Scholar Crossref - Vano-Galvan, Sergio, and Pedro Jaen. "Black hairy tongue." Cleveland Clinic journal of medicine, Vol. 75, No. 12, 2008, pp. 847-8.
Google Scholar Crossref - Rajarammohan, Karthik, et al. "Hairy Tongue-a series of 4 cases." Journal of Evolution of Medical and Dental Science, Vol. 9, No. 35, 2020, pp. 2567-71.
Google Scholar Crossref - Nisa, Lluís, and Roland Giger. "Black hairy tongue." The American journal of medicine, Vol. 124, No. 9, 2011, pp. 816-7.
Google Scholar Crossref - Gurvits, Grigoriy E., and Amy Tan. "Black hairy tongue syndrome." World Journal of Gastroenterology: WJG, Vol. 20, No. 31, 2014, p. 10845.
Google Scholar Crossref - Aryal, Eliz, M. Rajbhandari, and Sabina Bhattarai. "Lansoprazole–induce black hairy tongue-a case report." Nepal Journal of Dermatology, Venereology & Leprology, Vol. 15, No. 1, 2017, pp. 52-4.
Google Scholar - Jhaj, Ratinder, Pushp Raj Gour, and Dinesh Prasad Asati. "Black hairy tongue with a fixed dose combination of olanzapine and fluoxetine." Indian Journal of Pharmacology, Vol. 48, No. 3, 2016, p. 318.
Google Scholar Crossref - Kumar, Sowmya Hemantha. Hairy tongue: A case report and review of literature. International Journal of Current Research, Vol. 8, No. 7, 2016, pp. 35022-4.
Google Scholar - Black hairy tongue: predisposing factors, diagnosis, and treatment
- Vautier, G., and B. B. Scott. "A oneâ?week quadruple eradication regimen for Helicobacter pylori in routine clinical practice." Alimentary pharmacology & therapeutics, Vol. 11, No. 1, 1997, pp. 107-8.
Google Scholar Crossref - Bradshaw, D. J., and P. D. Marsh. "Analysis of pH–driven disruption of oral microbial communities in vitro." Caries research, Vol. 32, No. 6, 1998, pp. 456-62.
Google Scholar Crossref - Thompson, Dennis F., and Tiffany L. Kessler. "Drugâ?induced black hairy tongue." Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy, Vol. 30, No. 6, 2010, pp. 585-93.
Google Scholar Crossref - Manabe, Motomu, et al. "Architectural organization of filiform papillae in normal and black hairy tongue epithelium: dissection of differentiation pathways in a complex human epithelium according to their patterns of keratin expression." Archives of dermatology, Vol. 135, No. 2, 1999, pp. 177-81.
Google Scholar Crossref - Ren, Jing, et al. "Antibiotic-induced black hairy tongue: two case reports and a review of the literature." Journal of International Medical Research, Vol. 48, No. 10, 2020.
Google Scholar Crossref